Have you ever wondered how many electrons are in the HOMO, or the Highest Occupied Molecular Orbital? You might think this is a topic only for scientists in labs, but understanding it can unlock fascinating insights into the world around you.
From the way materials absorb light to the very essence of chemical reactions, the number of electrons in the HOMO plays a crucial role. As you dive into this article, you’ll discover how this seemingly complex concept is actually quite accessible and relevant to your everyday life.
So, if you’re curious about the unseen forces that shape the universe, keep reading to unravel the mystery of the electrons in the HOMO.
Homo In Molecular Orbitals
The Highest Occupied Molecular Orbital (HOMO) plays a crucial role in understanding molecules. It shows where the most energetic electrons are found. These electrons are important because they often take part in chemical reactions. The HOMO helps predict how molecules behave when they interact.
Definition Of Homo
The HOMO is the molecular orbital that contains the highest energy electrons. These electrons are still bound to the molecule but have more energy than others. The number of electrons in the HOMO depends on the molecule’s total electrons. It is the frontier orbital that can donate electrons during reactions.
Role In Chemical Reactions
The HOMO is key in many chemical processes. Electrons in the HOMO can move to other molecules’ orbitals. This transfer often starts chemical bonds or breaks them. Reactivity often depends on the energy level of the HOMO. A higher HOMO energy means electrons are more ready to react.
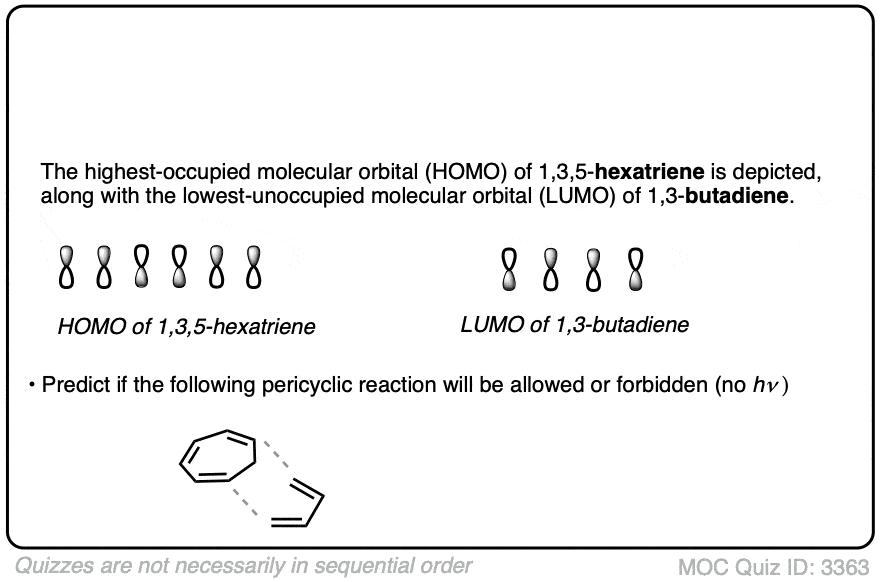
Credit: www.masterorganicchemistry.com
Electron Configuration Basics
Understanding electron configuration is key to grasping how electrons behave in molecules like the HOMO (Highest Occupied Molecular Orbital). It explains where electrons are most likely to be found and how they contribute to chemical bonding. Let’s break down the basics so you can follow the path electrons take from individual atoms to complex molecules.
Atomic Vs Molecular Orbitals
Atomic orbitals belong to single atoms, describing where electrons reside around a nucleus. Think of them as individual rooms inside an atom where electrons hang out.
Molecular orbitals, on the other hand, form when atoms combine to make molecules. These orbitals are like shared spaces, created by overlapping atomic orbitals, where electrons belong to the whole molecule, not just one atom.
Have you noticed how electrons don’t stay put in one spot but spread across atoms? This is why molecular orbitals are crucial—they explain electron behavior in bonds and molecules like HOMO.
Electron Filling Order
Electrons fill orbitals in a specific order based on energy levels, starting with the lowest energy first. This is called the Aufbau principle.
- Electrons fill the 1s orbital before moving to 2s.
- Orbitals within the same energy level fill in a sequence: s, then p, then d, then f.
- Each orbital can hold a maximum of two electrons with opposite spins.
Imagine filling seats in a theater from the front row backward; electrons prefer the lowest energy “seats” available. This order affects the properties of molecules and how the HOMO is formed.
Does this filling order change when atoms form molecules? Yes, because molecular orbitals can have different energy levels than atomic orbitals, altering where electrons settle.
Determining Electron Count In Homo
Understanding how many electrons reside in the Highest Occupied Molecular Orbital (HOMO) is crucial for grasping a molecule’s chemical behavior. The electron count in HOMO influences reactivity, stability, and interaction with other molecules. Pinpointing this number helps you predict how a molecule might respond in different environments.
Methods And Techniques
One common way to determine the electron count in HOMO is through computational chemistry software. Programs like Gaussian or ORCA calculate molecular orbitals and show electron distribution. These tools give you a clear visualization of where electrons are most likely to be found.
Another technique involves examining molecular orbital diagrams derived from spectroscopy data. Ultraviolet-visible (UV-Vis) spectroscopy, for example, can indicate electron transitions that reveal HOMO characteristics. This approach helps you connect experimental data with theoretical models.
Counting electrons also means knowing the molecule’s total number of valence electrons. From there, you fill molecular orbitals following the Aufbau principle, placing electrons in order of increasing energy. This step-by-step filling reveals exactly how many electrons occupy the HOMO.
Common Patterns In Molecules
Many organic molecules, like alkenes and aromatics, typically have two electrons in their HOMO. This is because molecular orbitals usually hold pairs of electrons. Recognizing this pattern simplifies predicting HOMO electron counts in similar compounds.
Transition metal complexes often show more complexity, with HOMO sometimes containing unpaired electrons. This variability can affect magnetism and reactivity. Have you noticed how these differences impact catalytic behavior in metals?
In conjugated systems, electrons delocalize over multiple atoms, which can raise the HOMO energy level and sometimes change the electron count. This delocalization makes molecules more reactive in certain chemical reactions. Understanding these patterns helps you anticipate molecular behavior more accurately.

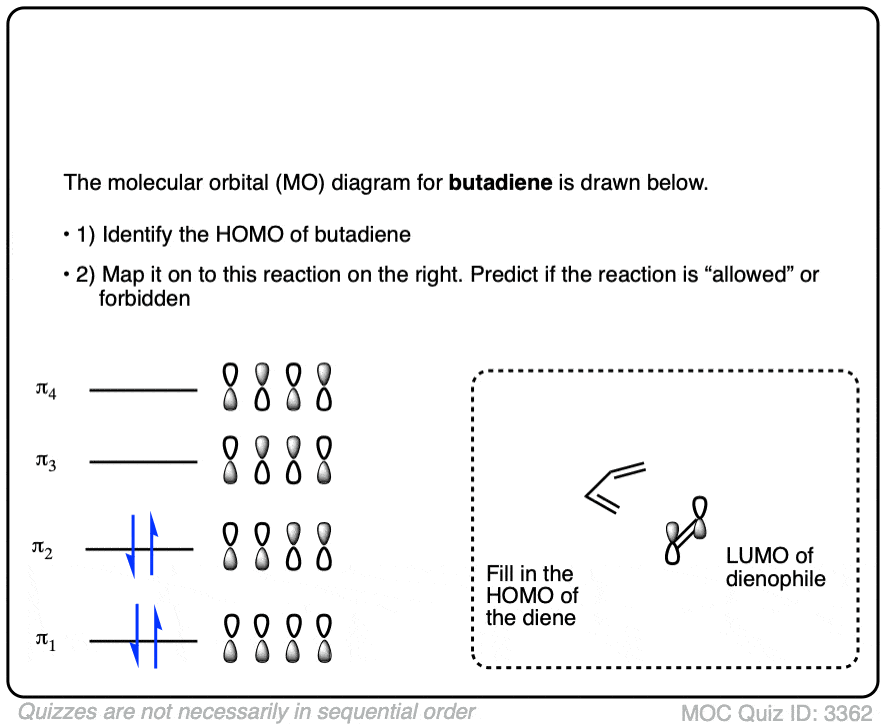
Credit: www.youtube.com
Significance Of Electron Number In Homo
The number of electrons in the Highest Occupied Molecular Orbital (HOMO) plays a crucial role in understanding chemical behavior. It helps predict how a molecule interacts with others and how it reacts in different environments. The electrons in the HOMO are the most ready to participate in chemical reactions because they are the outermost electrons.
Knowing the electron count in the HOMO gives insight into the molecule’s reactivity and its physical and chemical properties. This knowledge guides chemists in designing new compounds and materials with desired features.
Impact On Reactivity
The electrons in the HOMO are the first to react with other molecules. Their number determines how easily the molecule can donate electrons to another species.
- More electrons in the HOMO often mean higher reactivity.
- Fewer electrons may result in lower chemical activity.
- Electron-rich HOMOs tend to act as electron donors in reactions.
This makes the electron count vital for predicting reaction pathways and mechanisms.
Influence On Molecular Properties
The electron number in the HOMO affects a molecule’s color, magnetism, and electrical conductivity. These properties depend on how electrons are arranged and how many are available for bonding.
- Changes in HOMO electrons can alter absorption of light, affecting color.
- Magnetic behavior can be influenced by electron spin in the HOMO.
- Electrical conductivity depends on electron mobility in the HOMO.
Understanding these effects helps in the development of electronic devices and new materials.
Examples Across Different Molecules
Understanding how many electrons are in the Highest Occupied Molecular Orbital (HOMO) is key to predicting a molecule’s reactivity and stability. This varies widely among different types of molecules. Looking at examples helps you see the practical side of HOMO electron counts and how they influence chemical behavior.
Simple Diatomic Molecules
Take oxygen (O2) as a starting point. It has 12 electrons in its bonding and antibonding molecular orbitals, with the HOMO containing 2 electrons in the πantibonding orbitals. These two electrons explain why oxygen is paramagnetic and highly reactive.
In nitrogen (N2), the HOMO holds 2 electrons as well, but these electrons are in a bonding orbital, making nitrogen very stable and less reactive. This difference in HOMO electron placement can tell you why nitrogen is so inert compared to oxygen.
Hydrogen (H2) is even simpler. Its HOMO has 2 electrons in the bonding σ orbital, which is the only molecular orbital occupied. This simplicity makes it a great example of how HOMO electron count directly relates to bond strength.
Complex Organic Compounds
Now, consider benzene (C6H6). Its HOMO consists of 6 π electrons spread over the ring in delocalized orbitals. This delocalization stabilizes the molecule and affects how it reacts with electrophiles.
In molecules like acetone, the HOMO is mostly made up of nonbonding electrons on the oxygen atom. There are 2 electrons here, which explains why acetone can act as a nucleophile in many reactions.
What about larger molecules, like chlorophyll? Its HOMO has electrons primarily on the porphyrin ring system, enabling it to absorb light efficiently. This electron arrangement is why chlorophyll plays a crucial role in photosynthesis.
Looking at these examples, can you see how the number and location of electrons in the HOMO affect molecular properties? Understanding this can help you predict how molecules will behave in different chemical environments.
Credit: www.researchgate.net
Advanced Tools For Homo Analysis
Advanced tools help scientists study the Highest Occupied Molecular Orbital (HOMO) in detail. These tools reveal the number of electrons and their behavior in the HOMO. Understanding this helps predict chemical reactions and material properties. The following sections describe key techniques used for HOMO analysis.
Computational Chemistry Approaches
Computational chemistry uses computer models to simulate electron distribution. Methods like Density Functional Theory (DFT) calculate the number of electrons in the HOMO. These calculations provide visual maps of electron density and energy levels. Researchers adjust molecular structures in simulations to see how HOMO changes. These approaches are fast and cost-effective compared to experiments.
Common software tools include Gaussian, ORCA, and Spartan. They help predict HOMO characteristics for new molecules. Scientists use these models to design drugs, catalysts, and materials with specific properties. The accuracy depends on the chosen computational method and parameters.
Spectroscopic Methods
Spectroscopy measures how molecules interact with light. Techniques like Ultraviolet-Visible (UV-Vis) and Photoelectron Spectroscopy (PES) provide data on HOMO electrons. UV-Vis shows electron transitions from HOMO to higher orbitals. PES directly measures electron energy and counts electrons in the HOMO.
These experiments require specialized instruments and careful sample preparation. Results confirm computational predictions and give real-world data. Spectroscopy reveals changes in HOMO during chemical reactions. Scientists combine spectroscopic data with computations for better insights.
Frequently Asked Questions
How Many Electrons Are In The Homo Orbital?
The HOMO orbital contains the highest energy electrons in a molecule. Its electron count depends on the molecule’s total electrons and configuration.
Why Is Counting Electrons In Homo Important?
Knowing electron count in HOMO helps predict chemical reactivity. It shows how molecules interact during bonding or reactions.
How Does Homo Electron Count Affect Molecule Stability?
Higher electron density in HOMO can increase molecule reactivity. Stable molecules often have fully paired or lower-energy HOMO electrons.
Can Homo Electron Number Change In Excited States?
Yes, in excited states, electrons can move to higher orbitals. This changes the electron count in HOMO temporarily.
Conclusion
The number of electrons in the HOMO affects how molecules behave. It helps explain chemical reactions and stability. Understanding this concept makes studying chemistry easier. Keep exploring to learn more about molecular structures. Small details like electrons can reveal big secrets.
This knowledge builds a strong foundation for science learners. Stay curious and keep asking questions about atoms and electrons.
Leave a Reply