Have you ever wondered how your body turns the food you eat into energy while handling carbon dioxide (CO2)? The electron transport chain plays a key role in this process, but did you know it also has a connection to how CO2 is consumed?
Understanding how the electron transport chain interacts with CO2 can give you a clearer picture of your body’s energy system and its impact on your health. Keep reading, and you’ll discover the surprising ways this tiny molecular machine works hard to keep you energized and balanced.
Electron Transport Chain Basics
The electron transport chain (ETC) is a vital part of how cells produce energy. It works inside mitochondria, the cell’s power plants. The ETC helps turn the food we eat into usable energy.
This process uses electrons and oxygen to create a molecule called ATP. ATP stores energy for the cell to use. Understanding the basics of the ETC helps explain its role in consuming carbon dioxide (CO2).
Role In Cellular Respiration
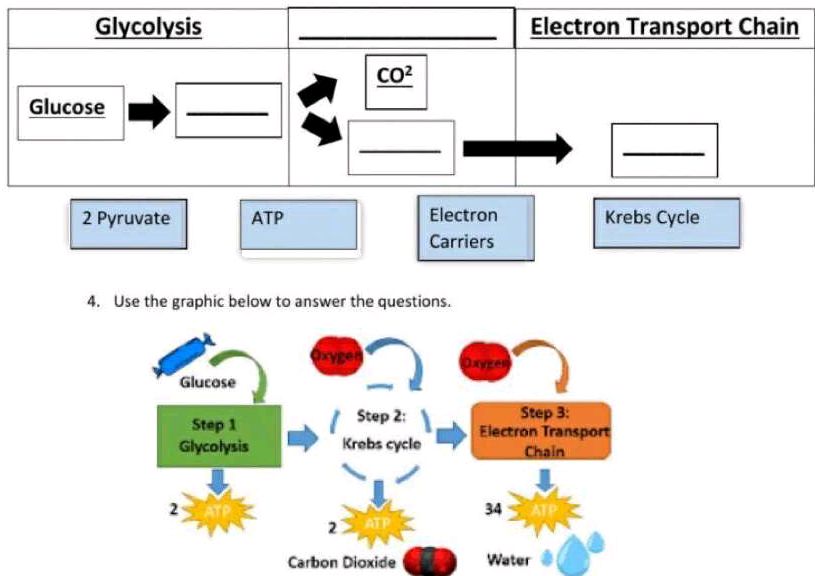
The ETC is the last stage of cellular respiration. It follows glycolysis and the Krebs cycle. Electrons from food molecules move through the chain.
As electrons pass through, the ETC pumps protons across the mitochondrial membrane. This creates a proton gradient. The energy from this gradient makes ATP.
Oxygen acts as the final electron acceptor. It combines with electrons and protons to form water. This step is crucial for the ETC to keep working.
Key Components
- Complex I (NADH Dehydrogenase):Accepts electrons from NADH and pumps protons.
- Complex II (Succinate Dehydrogenase):Transfers electrons from FADH2 without pumping protons.
- Complex III (Cytochrome bc1):Moves electrons and pumps protons across the membrane.
- Complex IV (Cytochrome c Oxidase):Transfers electrons to oxygen, forming water.
- ATP Synthase:Uses the proton gradient to produce ATP.

Credit: medium.com
Mechanisms Of Co2 Consumption
The mechanisms of CO2 consumption within the electron transport chain (ETC) reveal a fascinating interplay of biochemical reactions. Understanding these mechanisms helps clarify how cells manage carbon dioxide, a gas often seen only as a waste product. Let’s dive into how CO2 is actually used and transformed during these processes.
Understanding Co2’s Role
CO2 is not just a byproduct; it plays a subtle but important role in cellular respiration. In some pathways linked to the ETC, CO2 participates in reactions that help maintain the balance of molecules needed for energy production. Have you ever wondered how your cells keep this balance while you breathe out CO2?
In certain microorganisms and plant cells, CO2 consumption happens alongside the electron flow. This consumption helps drive the synthesis of key compounds, ensuring energy production continues smoothly. This means CO2 can be a resource, not just waste.
Chemical Reactions Involved
Several chemical reactions in the electron transport chain involve CO2, often through enzyme-driven steps. One example is the carboxylation reaction, where CO2 is added to molecules like phosphoenolpyruvate, helping regenerate intermediates needed for the cycle.
Another important reaction is the decarboxylation steps that release CO2 but are coupled with electron transfers. These reactions help create a proton gradient across membranes, which is essential for ATP synthesis.
- Carboxylation:Adding CO2 to organic molecules to form new compounds.
- Decarboxylation:Removing CO2 while transferring electrons to carriers like NADH.
- CO2 fixation:Some pathways fix CO2 into stable forms that feed back into metabolism.
Understanding these reactions can guide you in fields like bioengineering or environmental science, where controlling CO2 at the cellular level matters. What if we could enhance these natural processes to reduce atmospheric CO2? That’s a challenge worth exploring.
Biological Significance
The biological significance of the electron transport chain (ETC) extends beyond its well-known role in energy production. Understanding how it interacts with carbon dioxide (CO2) reveals insights into cellular metabolism and overall organism health. This interaction influences the efficiency of cellular processes and shapes how cells respond to environmental changes.
Impact On Cellular Efficiency
The electron transport chain indirectly influences CO2 consumption by driving metabolic reactions that fix or release CO2. Efficient ETC activity ensures that cells maintain a balanced energy supply, which is crucial for processes like the Calvin cycle in plants, where CO2 is fixed into organic molecules.
When the ETC operates optimally, it supports ATP production that powers enzymes responsible for CO2 assimilation. If the ETC slows down, cells may face energy shortages, reducing their ability to consume CO2 effectively. Have you ever noticed how your energy levels drop when your body is under stress? At the cellular level, something similar happens when the ETC is compromised.
Comparative Analysis With Other Pathways
Unlike pathways such as glycolysis or the Krebs cycle, which directly produce or consume CO2, the electron transport chain plays a supportive but critical role. It provides the energy needed for these pathways to function efficiently.
| Pathway | Role in CO2 Consumption | Energy Involvement |
|---|---|---|
| Electron Transport Chain | Supports CO2 fixation indirectly by supplying ATP and NADPH | High energy yield through oxidative phosphorylation |
| Calvin Cycle | Directly fixes CO2 into sugars | Consumes ATP and NADPH |
| Krebs Cycle | Releases CO2 during metabolism | Generates NADH and FADH2 for ETC |
Comparing these processes helps you see how the ETC is a powerhouse that fuels CO2-related metabolism rather than a direct consumer. This distinction is crucial for biotechnological applications aiming to reduce atmospheric CO2 by enhancing cellular pathways.

Credit: quizlet.com
Research And Discoveries
Research on how the electron transport chain interacts with CO2 has grown rapidly. Scientists explore the subtle ways this process may affect carbon dioxide levels. These discoveries reveal new insights into cellular energy and carbon cycles.
Studying this topic helps us understand energy production and its impact on the environment. Research bridges biology and chemistry to clarify how cells manage CO2 during respiration.
Recent Studies And Findings
Recent studies show that the electron transport chain primarily consumes oxygen, not CO2. Yet, CO2 plays an indirect role in cellular respiration. Some research suggests CO2 influences the enzyme activity within mitochondria.
Other findings highlight that CO2 can affect the pH balance in cells. This balance, in turn, impacts the efficiency of the electron transport chain. Scientists have also found certain proteins that may interact with CO2 molecules.
- CO2 indirectly affects enzyme reactions in mitochondria
- Cellular pH changes influence electron transport chain efficiency
- Proteins may bind CO2 during cellular respiration
Technological Advances In Study
New technology allows researchers to observe the electron transport chain in real time. Advanced imaging techniques reveal how molecules move and interact in mitochondria. These tools provide clearer pictures of CO2’s role.
Mass spectrometry helps detect tiny changes in carbon compounds. This method tracks how CO2 enters and leaves cells during respiration. Computational models simulate electron transport and CO2 dynamics.
| Technology | Purpose |
|---|---|
| Advanced Microscopy | Visualize mitochondria and molecule movement |
| Mass Spectrometry | Measure carbon compound changes |
| Computational Models | Simulate electron transport and CO2 interaction |
Implications For Biotechnology
The electron transport chain (ETC) is traditionally known for its role in energy production within cells. However, its connection to CO2 consumption presents exciting opportunities for biotechnology. Understanding these implications can open new doors for sustainable technologies and innovative bio-based solutions.
Applications In Carbon Capture
Using the ETC to consume CO2 offers a biological route for carbon capture. Unlike mechanical methods, this approach can be more energy-efficient and environmentally friendly. You could imagine designing microbes that naturally pull CO2 from the air or industrial emissions and convert it into useful products.
This biological carbon capture could reduce reliance on chemical scrubbers and high-energy processes. It’s a promising direction for industries looking to cut their carbon footprint without massive infrastructure changes. How might your industry benefit from integrating such living systems for CO2 management?
Potential For Bioengineering
Bioengineering the electron transport chain to enhance CO2 consumption is a growing field. Scientists are tweaking the proteins and pathways involved to increase efficiency and output. You could participate in creating microbes tailored to specific environments or tasks, such as producing biofuels or biodegradable plastics from captured CO2.
Imagine customizing bacteria that not only consume CO2 but also generate valuable compounds as byproducts. This dual function could make your projects more sustainable and cost-effective. What kinds of products would you want to create if you had control over these biological systems?
Challenges And Controversies
The electron transport chain (ETC) plays a crucial role in cellular respiration. Yet, how it interacts with CO2 remains a topic full of challenges and debates. Scientists face many difficulties in fully explaining CO2 consumption by the ETC. This section explores key controversies and gaps in knowledge.
Debates In Scientific Community
Some researchers argue that the ETC directly consumes CO2 during its process. Others believe CO2 involvement is indirect or minimal. Experiments show varied results, causing disagreements on CO2’s exact role.
There is also debate about the enzymes involved. Identifying specific proteins that handle CO2 is difficult. The complexity of the ETC makes it hard to isolate these factors.
Scientists discuss how environmental conditions affect CO2 use. Differences in oxygen levels and pH may change how the ETC works with CO2. These variables add to the ongoing discussions.
Limitations Of Current Understanding
Current models of the ETC do not fully include CO2 consumption. Many studies focus on oxygen as the main electron acceptor, leaving CO2’s role unclear.
Technological limits restrict detailed observation inside mitochondria. This makes it hard to track CO2 during electron transport.
Research often relies on indirect measurements. This can lead to assumptions that may not capture the full picture.
More advanced tools and methods are needed. Better data will help clarify how the ETC interacts with CO2.

Credit: quizlet.com
Frequently Asked Questions
How Does The Electron Transport Chain Use Co2?
The electron transport chain (ETC) itself does not directly consume CO2. CO2 consumption occurs primarily during the Calvin cycle in photosynthesis, where CO2 is fixed into organic molecules. The ETC produces ATP and NADPH, which provide energy and reducing power for CO2 fixation.
Does The Electron Transport Chain Produce Or Consume Co2?
The electron transport chain neither produces nor consumes CO2 directly. Its main role is to generate ATP and NADPH by transferring electrons. CO2 involvement happens in connected processes like the Calvin cycle, not within the ETC itself.
What Role Does Co2 Play In The Electron Transport Chain?
CO2 does not play a direct role in the electron transport chain. Instead, CO2 is fixed in the Calvin cycle using ATP and NADPH produced by the ETC during photosynthesis.
Can The Electron Transport Chain Reduce Co2 Levels?
The electron transport chain indirectly supports CO2 reduction by generating energy carriers. These carriers drive CO2 fixation in the Calvin cycle, thus helping lower CO2 levels in photosynthetic organisms.
Conclusion
The electron transport chain plays a key role in energy production. It does not directly consume CO2 but helps produce energy needed for carbon fixation. Cells use this energy to convert CO2 into useful molecules during photosynthesis or other processes.
Understanding this helps explain how life transforms carbon in nature. This process supports all living things by making energy available. Knowing how the electron transport chain works gives insight into the balance of gases on Earth. It shows the link between energy flow and carbon cycling clearly.
Leave a Reply