Are you curious about the mysterious world of chemistry and how certain groups of atoms can influence powerful reactions? If you’ve ever wondered how electron donating groups can alter the behavior of dienes, you’re in the right place.
Understanding the effect on the Highest Occupied Molecular Orbital (HOMO) of a diene isn’t just for the chemistry enthusiasts; it opens a window into the fascinating interactions that underpin everything from industrial processes to the natural world. By the end of this article, you’ll discover how these groups can tweak and transform the very nature of chemical reactions, potentially changing the way you perceive molecular chemistry.
Dive in and uncover the secrets that could transform your understanding of the microscopic world!
Basics Of Electron Donating Groups
Electron donating groups (EDGs) play an important role in chemistry. They influence how molecules behave, especially dienes. EDGs push electrons toward the molecule’s core. This changes the energy levels of molecular orbitals. Understanding EDGs helps explain many chemical reactions and properties.
In dienes, EDGs affect the Highest Occupied Molecular Orbital (HOMO). This change alters how dienes react with other chemicals. The basics of electron donating groups reveal why this happens.
Types Of Electron Donating Groups
Electron donating groups can be classified by how they release electrons. The two main types are:
- Alkyl Groups:These groups release electrons through their sigma bonds. Examples include methyl (-CH3) and ethyl (-C2H5).
- Groups with Lone Pairs:These have atoms like oxygen or nitrogen with lone electron pairs. Examples are hydroxyl (-OH), amino (-NH2), and alkoxy (-OR) groups.
Each type affects the molecule differently based on its electron releasing power.
Mechanism Of Electron Donation
Electron donating groups add electron density to the molecule. They do this mainly by two ways:
- Inductive Effect:EDGs push electrons through sigma bonds. This effect is weaker but spreads across the molecule.
- Resonance Effect:EDGs with lone pairs share electrons through pi bonds. This effect is stronger and more direct.
In dienes, resonance is especially important. It raises the energy of the HOMO. This makes dienes more reactive in many cases.
Credit: www.masterorganicchemistry.com
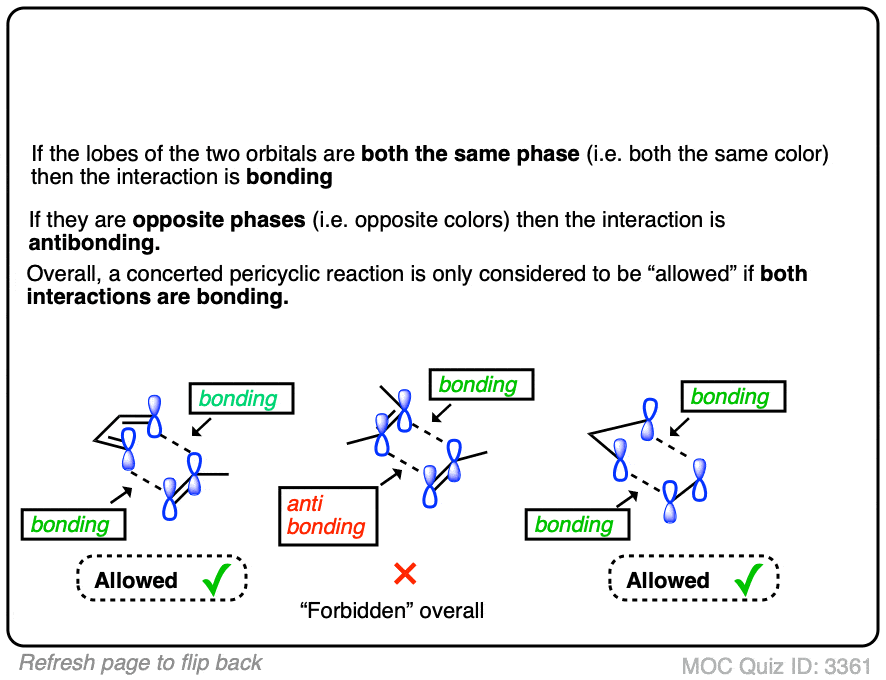
Homo In Dienes
The Highest Occupied Molecular Orbital (HOMO) plays a crucial role in determining the chemical behavior of dienes. Understanding how the HOMO interacts with other molecules can help you predict reactivity patterns, especially in reactions like Diels-Alder cycloadditions. The energy level and shape of the HOMO directly influence how dienes respond to electron donating groups.
Role Of Homo In Diene Reactivity
The HOMO is essentially the electron-rich area of the diene that participates in bonding during reactions. When you look at diene reactivity, the HOMO donates electrons to the Lowest Unoccupied Molecular Orbital (LUMO) of an electrophile. This electron donation drives the formation of new bonds and determines how fast and efficiently the reaction proceeds.
For example, a diene with a higher energy HOMO will react more readily because its electrons are more available for bonding. Have you ever noticed how some dienes react faster under the same conditions? The HOMO energy level often explains this difference.
Factors Influencing Homo Energy Levels
Several factors affect the energy of the HOMO in dienes. One major factor is the presence of electron donating groups (EDGs), which push electron density into the diene system, raising the HOMO energy. This makes the diene more reactive as it becomes easier to donate electrons.
- Type of substituent:Alkyl or alkoxy groups increase HOMO energy more than halogens.
- Position of substitution:Substituents directly attached to the diene carbons have a stronger effect than those further away.
- Conjugation:Extended conjugation can delocalize electron density, affecting HOMO energy and shape.
Imagine tweaking your diene by adding an -OH group versus a methyl group. The -OH, being a stronger electron donor, raises the HOMO energy more significantly, which can dramatically change how the molecule reacts. Have you tried experimenting with different substituents to see their effect firsthand? It’s a straightforward way to connect theory with practical results.
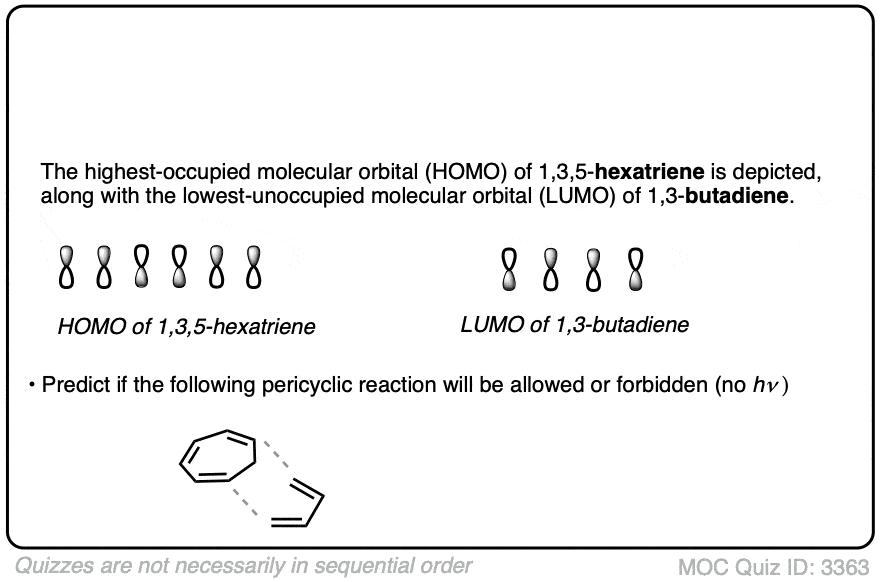
Impact Of Electron Donating Groups On Homo
The presence of electron donating groups (EDGs) on a diene has a significant effect on its Highest Occupied Molecular Orbital (HOMO). These groups alter the electronic environment, which in turn influences the energy and reactivity of the diene. Understanding this impact helps you predict how a diene will behave in chemical reactions, especially in processes like Diels-Alder reactions.
Energy Level Modulation
Electron donating groups push electron density into the diene’s conjugated system. This addition raises the energy level of the HOMO, making it more reactive. You might wonder why this matters—higher HOMO energy means the diene can more easily donate electrons during bonding.
For example, when a methoxy (-OCH3) group is attached, the HOMO energy increases noticeably compared to an unsubstituted diene. This change can speed up reactions where the diene acts as an electron donor. Have you considered how tweaking substituents on your diene could speed up or slow down your reactions?
Orbital Interaction Changes
Electron donating groups don’t just raise the energy; they also reshape the shape and symmetry of the HOMO. This affects how well the diene’s orbitals overlap with those of other molecules during reactions. Better overlap can mean stronger bonds and more efficient reactions.
Take the case of an amino (-NH2) substituent. Its lone pair interacts with the diene’s π system, changing the distribution of electron density in the HOMO. This can improve orbital alignment with the dienophile, increasing reaction rates and selectivity.
Have you noticed how subtle changes in substituents can lead to big differences in reaction outcomes? Paying attention to these orbital interactions could give you greater control over your synthetic strategies.

Credit: en.wikipedia.org
Consequences For Diene Reactivity
Electron donating groups (EDGs) change the way dienes behave in chemical reactions. They affect the highest occupied molecular orbital (HOMO) of the diene. This change in the HOMO energy impacts the diene’s reactivity. EDGs increase electron density, making the diene more reactive. Understanding this effect helps in predicting and controlling reactions involving dienes.
Effect On Diels-alder Reactions
EDGs raise the energy level of the diene’s HOMO. A higher HOMO energy means the diene can react more easily with dienophiles. This speeds up Diels-Alder reactions. The diene becomes more nucleophilic and reactive. The reaction often needs less heat or catalyst. This effect can improve yields and selectivity in the synthesis of complex molecules.
Influence On Polymerization Processes
In polymerization, EDGs also raise the HOMO energy of dienes. This change makes dienes more reactive towards polymerization initiators. The polymer chains grow faster and with better control. EDGs can affect the type and properties of the resulting polymer. This influence helps create polymers with specific characteristics and improved performance.
Experimental Evidence And Studies
Experimental evidence plays a crucial role in understanding how electron donating groups affect the HOMO of dienes. Researchers use various techniques to observe changes in electronic structure and reactivity. These studies help confirm theoretical predictions and provide deeper insights into molecular behavior.
Spectroscopic Observations
Spectroscopy offers direct evidence of electronic changes in dienes with electron donating groups. UV-Vis spectroscopy shows shifts in absorption peaks, indicating altered HOMO energy levels. Electron donating groups often cause bathochromic shifts, meaning absorption moves to longer wavelengths.
NMR spectroscopy reveals changes in chemical environments near the diene. Electron donating groups increase electron density, causing upfield shifts in proton signals. These shifts confirm increased electron availability at the HOMO.
Infrared spectroscopy detects changes in bond vibrations. Electron donating groups weaken certain bonds by increasing electron density, changing vibrational frequencies. These variations align with HOMO modification theories.
Computational Chemistry Insights
Computational studies complement experiments by simulating electronic structures. Quantum chemical calculations map HOMO orbitals and their energies accurately. Electron donating groups consistently raise HOMO energy, making dienes more reactive.
Density Functional Theory (DFT) is popular for these simulations. DFT results match spectroscopic data closely, strengthening conclusions. Visualizing HOMO orbitals helps understand electron distribution changes due to substituents.
Computational models also predict reactivity trends. Higher HOMO energy often means increased nucleophilicity. These insights guide chemists in designing dienes with desired properties.
Designing Dienes With Tailored Properties
Designing dienes with tailored properties is essential in many chemical and material science fields. Electron donating groups (EDGs) play a crucial role in modifying the highest occupied molecular orbital (HOMO) of dienes. By changing the HOMO energy, EDGs influence the diene’s reactivity and stability. This control enables chemists to create dienes with specific behaviors for various applications.
Selecting Suitable Electron Donating Groups
Choosing the right electron donating group is key to tuning diene properties. Common EDGs include alkyl, alkoxy, and amino groups. These groups increase electron density in the diene, raising the HOMO level. A higher HOMO often means better reactivity in cycloaddition reactions. The strength and position of the EDG affect how much the HOMO shifts. Proper selection balances stability and reactivity for the intended use.
Applications In Material Science
Tailored dienes find wide use in material science. Modified HOMO levels improve polymerization processes. This leads to polymers with desired flexibility or strength. EDG-substituted dienes also enhance light absorption in organic electronics. Their adjusted electronic properties support better performance in solar cells and sensors. Designing dienes with specific EDGs helps create advanced materials with precise functions.
Credit: www.masterorganicchemistry.com
Frequently Asked Questions
What Is An Electron Donating Group In Dienes?
An electron donating group (EDG) is a substituent that pushes electrons toward the diene system. This increases electron density on the diene. EDGs stabilize the highest occupied molecular orbital (HOMO) and affect the diene’s reactivity in chemical reactions.
How Do Edgs Affect The Homo Energy Of Dienes?
EDGs raise the energy level of the HOMO in dienes. This makes the diene more nucleophilic and reactive. Higher HOMO energy facilitates easier overlap with electrophiles, enhancing reaction rates in processes like Diels-Alder reactions.
Why Is Homo Important For Diene Reactivity?
The HOMO controls how dienes interact with electrophiles. A higher HOMO energy means better electron donation capability. This directly influences the diene’s ability to undergo cycloaddition and other reactions efficiently.
Can Edgs Change Diene Reaction Selectivity?
Yes, EDGs influence regioselectivity and stereoselectivity in diene reactions. By altering electron density, EDGs direct where and how the diene reacts. This helps in designing specific products in organic synthesis.
Conclusion
Electron donating groups raise the HOMO energy of dienes. This change makes dienes more reactive in chemical reactions. It helps in understanding how molecules behave during synthesis. Knowing this effect guides chemists in designing better compounds. Simple changes in groups cause big shifts in reactivity.
This knowledge is key for many fields like pharmaceuticals and materials science. Studying electron donating groups opens doors to improved chemical processes. The relationship between structure and reactivity becomes clearer. Understanding this topic helps build a strong foundation in organic chemistry.
Leave a Reply