Do you ever wonder about the tiny particles that make up the world around you? If you’re curious about chemistry, you might find yourself asking questions like, “How many electrons are present in 1,3,5-hexatriene homo?”
You’re not alone. This question might seem complex, but it’s fascinating when you break it down. Understanding this can unlock a new level of appreciation for molecular structures and their behaviors. Imagine being able to grasp the intricacies of how these electrons interact, shaping the characteristics of this intriguing compound.
By the end of this article, you’ll not only know the answer but also gain insights into why this knowledge matters. So, let’s dive in and explore the microscopic world that holds immense power over the nature of substances.

Credit: medium.com
Basics Of 1,3,5-hexatriene
Understanding the basics of 1,3,5-hexatriene helps clarify its electron count and behavior. This organic compound consists of a chain of six carbon atoms with alternating double bonds. Its structure plays a key role in its chemical properties and reactions.
Molecular Structure
1,3,5-Hexatriene has six carbon atoms linked in a straight chain. Three double bonds alternate with single bonds between them. Each carbon atom bonds with hydrogen atoms to complete its valence. This pattern creates a conjugated system of pi electrons across the molecule.
- Six carbon atoms in a linear chain
- Three alternating double bonds at positions 1, 3, and 5
- Hydrogen atoms fill the remaining valences
- Conjugation allows electron delocalization
Bonding In Hexatriene
The bonding involves sigma and pi bonds. Sigma bonds connect the carbon atoms and hydrogens strongly. Pi bonds form above and below the carbon chain where double bonds exist. These pi electrons are shared across the molecule, creating a conjugated system. This conjugation affects the molecule’s stability and electron distribution.
- Sigma bonds hold the main structure
- Pi bonds create regions of electron density
- Conjugated pi bonds allow electrons to move freely
- Electron delocalization stabilizes the molecule
Electron Count In Hexatriene
Understanding the electron count in 1,3,5-hexatriene helps grasp its chemical behavior. This molecule has a chain of six carbon atoms with alternating double bonds. Electrons in hexatriene play key roles in bonding and reactivity. Knowing the types and numbers of electrons reveals how the molecule forms and reacts.
Types Of Electrons Present
Hexatriene contains two main types of electrons. These are sigma (σ) electrons and pi (π) electrons. Sigma electrons form strong single bonds between atoms. Pi electrons are found in double bonds and create areas of electron density above and below the molecule’s plane. Both types influence hexatriene’s structure and properties.
Sigma And Pi Electrons
Each carbon-carbon single bond has two sigma electrons. Each double bond contains two sigma electrons plus two pi electrons. In 1,3,5-hexatriene, there are three double bonds along the chain. This means the molecule has:
- Seven sigma bonds (six C-C and one C-H bond per carbon)
- Three pi bonds from the three double bonds
Counting electrons:
- Sigma electrons = 14 (from 7 sigma bonds × 2 electrons each)
- Pi electrons = 6 (from 3 pi bonds × 2 electrons each)
Total electrons involved in bonding equal 20. This electron distribution affects hexatriene’s stability and how it absorbs light. The pi electrons are especially important in reactions and electronic transitions.
Homo In 1,3,5-hexatriene
The HOMO in 1,3,5-hexatriene plays a crucial role in its chemical behavior. It represents the highest energy level occupied by electrons. Understanding the HOMO helps explain how this molecule interacts with light and other chemicals. This section breaks down the HOMO concept and its electron configuration in 1,3,5-hexatriene.
Definition Of Homo
HOMO stands for Highest Occupied Molecular Orbital. It is the topmost energy level that contains electrons in a molecule. Electrons in the HOMO can move to higher energy states when energized. This orbital defines many chemical properties, such as reactivity and bonding. In 1,3,5-hexatriene, the HOMO involves pi electrons from the conjugated double bonds.
Homo Electron Configuration
1,3,5-Hexatriene has six pi electrons from its three double bonds. These electrons fill molecular orbitals from lowest to highest energy. The HOMO contains the highest energy pi electrons that are still occupied. For 1,3,5-hexatriene, the HOMO holds two electrons. These electrons are paired and occupy a bonding molecular orbital formed by the overlap of p orbitals. This arrangement stabilizes the molecule and allows it to absorb specific wavelengths of light.
Counting Electrons In Homo
Counting electrons in the Highest Occupied Molecular Orbital (HOMO) of 1,3,5-hexatriene is key to understanding its chemical behavior. The HOMO holds electrons that are most ready to participate in reactions. Knowing exactly how many electrons are in this orbital gives you insight into the molecule’s reactivity and stability.
Role Of Pi Electrons
The pi electrons come from the double bonds in 1,3,5-hexatriene. There are three double bonds, each contributing two pi electrons, making a total of six pi electrons. These electrons are not localized; instead, they are spread out over the conjugated system.
These delocalized pi electrons form the molecular orbitals, including the HOMO. You can think of the HOMO as the highest energy level that is still filled with electrons. These pi electrons in the HOMO are the ones that will most likely interact during chemical reactions.
Electron Distribution In Homo
In 1,3,5-hexatriene, the HOMO consists of six pi electrons spread across the conjugated chain. These electrons occupy molecular orbitals formed by the combination of p orbitals on each carbon in the chain.
The electron distribution in the HOMO is such that the electrons are delocalized, which stabilizes the molecule. This delocalization means the electrons are shared over several atoms, not just confined to one double bond.
Do you realize how this electron sharing affects reaction pathways? The delocalized electrons in the HOMO make the molecule more reactive at certain positions, influencing where new bonds might form.
Significance Of Electron Count
Understanding the electron count in 1,3,5-hexatriene homo is more than just a number game. It reveals how the molecule behaves and interacts at the most fundamental level. Knowing the exact number of electrons helps you predict its stability and reactivity, which is crucial for designing chemical reactions or new materials.
Chemical Reactivity
The electron count directly influences how 1,3,5-hexatriene homo reacts with other substances. Electrons in the molecule’s conjugated system create areas of high electron density, making it prone to certain types of chemical attacks.
For example, these electrons can participate in addition reactions, where new atoms or groups attach to the molecule. If you understand the electron distribution, you can predict which parts of the molecule are more reactive and how to control the outcome of a reaction.
Molecular Orbital Theory Application
Electron count is essential when applying molecular orbital theory to 1,3,5-hexatriene homo. This theory explains how atomic orbitals combine to form molecular orbitals, which hold the electrons.
By counting the electrons, you can fill these orbitals and determine the molecule’s energy state. This helps you understand whether the molecule is in a stable or excited state and how it might absorb or emit light—a key factor in photochemistry and materials science.
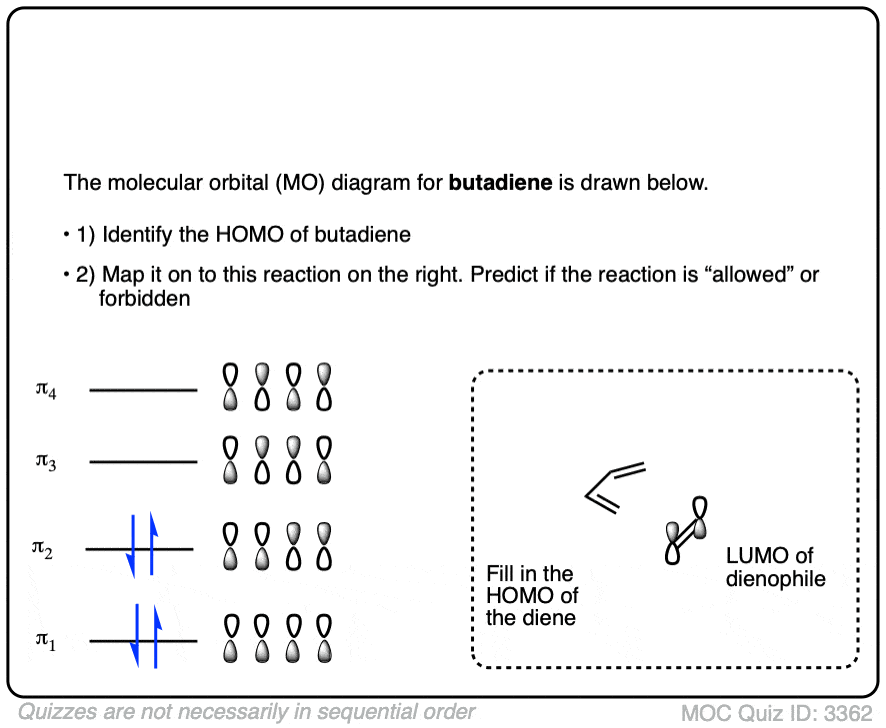
Credit: www.masterorganicchemistry.com
Experimental And Computational Approaches
Understanding how many electrons are present in 1,3,5-hexatriene HOMO requires a combination of experimental and computational approaches. These methods provide complementary insights—experiments reveal real-world data, while computations offer detailed electronic structure analysis. Together, they give a clearer picture of the molecule’s electronic behavior.
Spectroscopic Methods
Spectroscopy allows you to observe the energy levels of electrons directly. Techniques like UV-Vis and photoelectron spectroscopy help identify the highest occupied molecular orbital (HOMO) by measuring electron transitions.
Photoelectron spectroscopy, for example, ejects electrons from the molecule and measures their energies. This tells you which electrons are in the HOMO and how many there are. Using spectroscopy, you get experimental evidence of electron distribution without guessing.
Quantum Chemical Calculations
Computational chemistry uses quantum mechanics to predict electron counts and distributions in molecules like 1,3,5-hexatriene. Software programs calculate molecular orbitals and energies, showing you the HOMO’s electron population.
These calculations often confirm or refine what spectroscopy suggests. You can visualize electron clouds, check orbital shapes, and even simulate how electrons behave during reactions. This approach gives you precise numbers on electron occupancy, enhancing your understanding beyond what experiments alone provide.
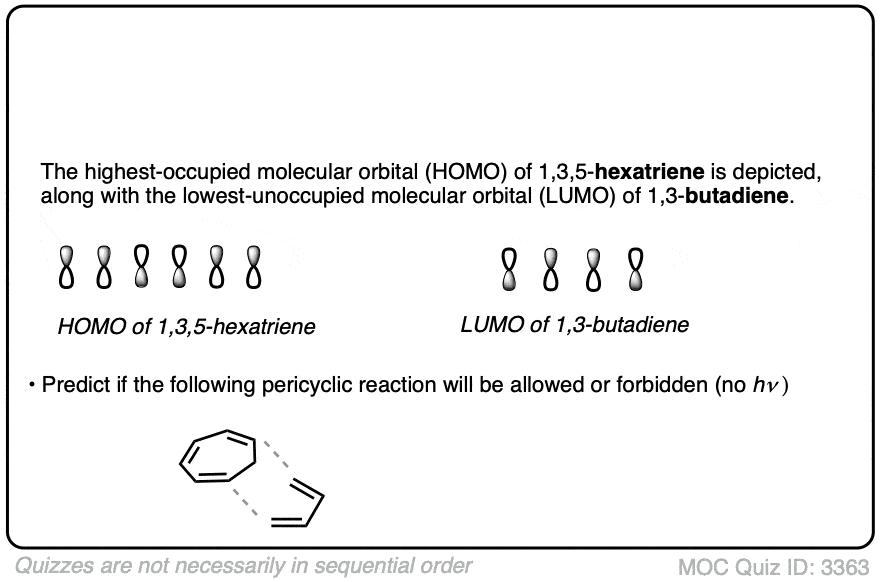
Credit: www.masterorganicchemistry.com
Frequently Asked Questions
How Many Electrons Are In 1,3,5-hexatriene Homo?
The HOMO of 1,3,5-hexatriene contains six π electrons. These electrons occupy the highest energy molecular orbital in the conjugated system. The HOMO reflects the molecule’s electron-rich region, influencing its chemical reactivity and properties.
What Defines The Homo In 1,3,5-hexatriene?
The HOMO is the highest occupied molecular orbital with electrons in 1,3,5-hexatriene. It consists mainly of π-bonding electrons in the conjugated system. This orbital determines how the molecule interacts with electrophiles or absorbs light.
Why Is Homo Important For 1,3,5-hexatriene?
HOMO controls the molecule’s chemical reactivity and optical properties. It shows where electrons are most available for reactions. Understanding HOMO helps predict behavior in organic synthesis and photochemistry.
How Are Electrons Distributed In 1,3,5-hexatriene’s Homo?
Electrons in the HOMO are delocalized over the conjugated double bonds. This delocalization stabilizes the molecule and lowers its energy. It allows efficient overlap of p-orbitals across the hexatriene chain.
Conclusion
1,3,5-Hexatriene has 18 electrons in its HOMO. These electrons play a key role in its chemical behavior. Understanding this helps predict reactions and properties. The HOMO shows where electrons are most reactive. This knowledge supports studies in organic chemistry and materials science.
Simple electron counts can reveal much about molecules. Keep exploring electron arrangements to learn more.
Leave a Reply