Have you ever wondered what makes up an electron? You might think it’s just a tiny particle, but there’s more to it than meets the eye.
Understanding what an electron is made of can change the way you see the building blocks of everything around you. You’ll discover the fascinating details behind this fundamental part of matter. Get ready to unlock the secrets of the electron and see how it shapes the world you live in.
Keep reading—you’ll be surprised by what you learn!
Credit: informatica-educativa-i-upn.fandom.com
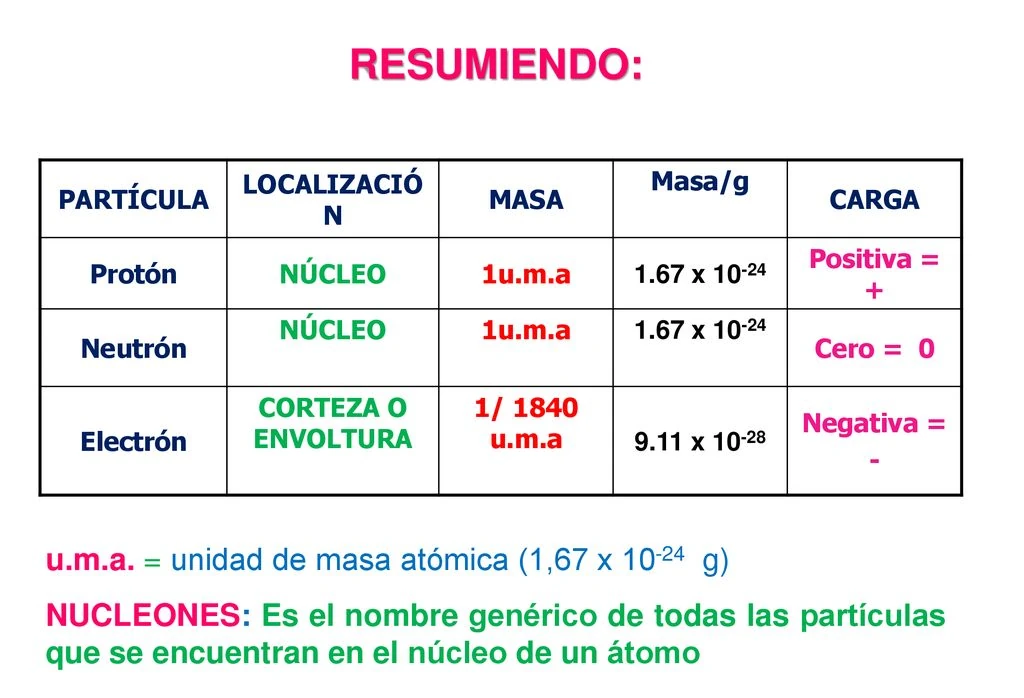
Nature Of Electrons
Electrons are fundamental particles that play a crucial role in the structure of atoms and the behavior of matter. Understanding their nature helps you grasp how everything around you functions, from electricity to chemical reactions. Let’s uncover what makes electrons unique and essential.
What Are Electrons Made Of?
Electrons are elementary particles, meaning they are not made up of smaller components. Unlike protons and neutrons, which consist of quarks, electrons stand alone as indivisible units. This simplicity makes them one of the building blocks of the universe.
Properties That Define Electrons
- Charge:Electrons carry a negative electric charge, exactly opposite to the positive charge of protons.
- Mass:Their mass is extremely small—about 1/1836 the mass of a proton.
- Spin:Electrons have an intrinsic property called spin, which contributes to magnetic effects.
These properties influence how electrons interact with other particles and fields, shaping the physical world.
Wave-particle Duality Of Electrons
You might think of electrons as tiny balls, but they also behave like waves. This dual nature means electrons can spread out and interfere with themselves, which is why they don’t simply orbit the nucleus like planets around the sun. This behavior is key to technologies like electron microscopes and semiconductors.
How Electrons Affect Your Daily Life
Every time you switch on a light or use your phone, electrons are at work. Their movement through wires creates electricity, powering your devices. Understanding their nature can help you appreciate how modern technology depends on these tiny particles.
Questions To Ponder
- How does knowing that electrons are fundamental particles change your view of matter?
- What would happen if electrons had a different charge or mass?
- Can you think of everyday devices that rely directly on the unique properties of electrons?

Credit: quimica4toescuelatecnica.wordpress.com
Fundamental Particles
Fundamental particles form the building blocks of everything in the universe. They are the smallest pieces that make up matter. Understanding these particles helps us learn what electrons are made of. Electrons belong to a group called fundamental particles because they have no smaller parts inside. These particles follow specific rules and behave in unique ways.
Quarks And Leptons
Quarks and leptons are two main types of fundamental particles. Quarks combine to form protons and neutrons. Leptons include electrons, muons, and neutrinos. Electrons are a type of lepton. Unlike quarks, electrons do not break down into smaller parts. They have a fixed electric charge and a very small mass.
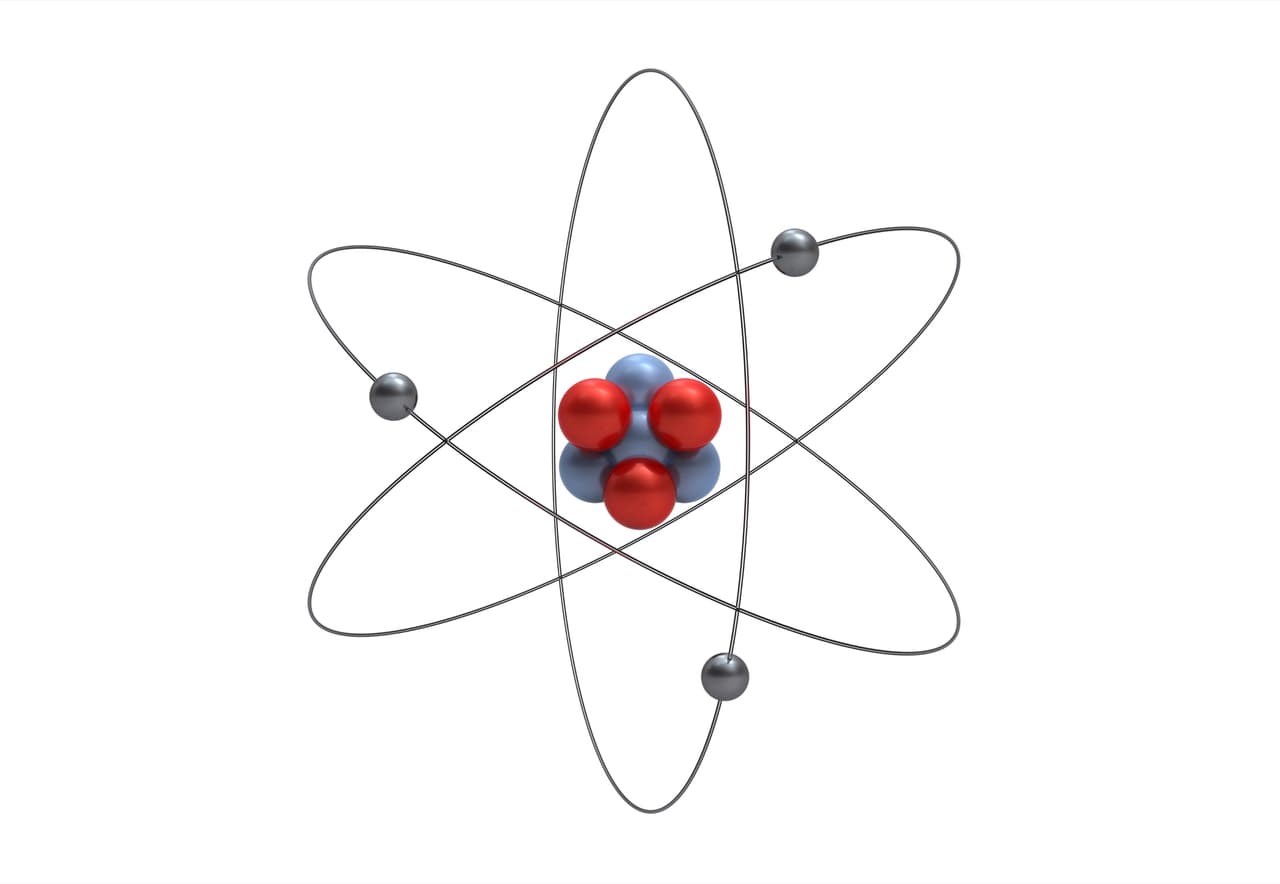
Role Of Electrons In Atoms
Electrons orbit the nucleus of an atom. They create a cloud of negative charge around the nucleus. This cloud balances the positive charge of protons inside the nucleus. Electrons determine how atoms bond and react with other atoms. They play a key role in electricity and chemical reactions. Without electrons, atoms could not form molecules or matter as we know it.
Quantum Properties
Quantum properties define the strange and fascinating behavior of electrons. These tiny particles do not behave like everyday objects. Instead, they follow the rules of quantum mechanics. Understanding these properties helps us learn how electrons exist and move.
Wave-particle Duality
Electrons act both as particles and waves. This dual nature means they can collide like tiny balls or spread out like waves. It explains many strange effects in experiments. For example, electrons can create patterns of light and dark bands, similar to waves on water. This duality is key to modern physics and technology.
Uncertainty Principle
The uncertainty principle states that we cannot know both an electron’s position and speed exactly. The more precisely we measure one, the less we know about the other. This idea shows the limits of measuring tiny particles. It means electrons do not have fixed paths like planets. Instead, their position and motion are described by probabilities.
Electron Interactions
Electron interactions shape much of the behavior of matter. These tiny particles do not exist in isolation. They constantly interact with forces and fields around them. Understanding these interactions helps explain electricity, magnetism, and chemical reactions.
Electromagnetic Forces
Electrons carry a negative electric charge. This charge causes them to attract or repel other charged particles. Electromagnetic forces guide how electrons move and interact.
These forces keep electrons bound to atoms. They also allow electrons to flow in wires, creating electric currents. The strength of electromagnetic force is much stronger than gravity at small scales.
Spin And Magnetism
Electrons have a property called spin. Spin is a type of intrinsic angular momentum. It behaves like a tiny magnet inside the electron.
Spin causes electrons to align with magnetic fields. This alignment creates magnetic effects in materials. Electron spin plays a key role in technologies like MRI and hard drives.
Electron Configuration
Understanding electron configuration is key to grasping what an electron is made of and how it behaves within an atom. It describes how electrons are arranged around the nucleus, which directly impacts chemical properties and reactions. The way electrons fill different energy levels and orbitals shapes the entire structure of matter.
Energy Levels
Electrons occupy specific energy levels, often called shells, surrounding the nucleus. These levels are numbered starting from 1, closest to the nucleus, and increase as you move outward. Each energy level can hold a limited number of electrons, influencing how atoms bond and interact.
Think about how your phone battery charges in stages. Similarly, electrons fill lower energy levels before moving to higher ones. This arrangement keeps the atom stable and balanced.
Orbitals And Shells
Within each energy level, electrons are found in orbitals—regions where they are most likely to be. Orbitals come in different shapes: s (sphere), p (dumbbell), d, and f, each with a distinct capacity for electrons.
For example, the first shell has just one s orbital and can hold up to 2 electrons. The second shell has one s and three p orbitals, holding up to 8 electrons in total. This pattern continues, explaining why elements in the same column of the periodic table often share similar properties.
Have you ever wondered why some elements are more reactive than others? The answer often lies in their electron configuration, especially the arrangement in the outermost orbitals. Understanding this can help you predict chemical behavior with confidence.
Modern Discoveries
Modern discoveries have deepened our understanding of what composes an electron. Scientists now explore beyond the basic idea of electrons as simple particles. These findings reveal complex behavior and properties at the smallest scales. The electron is no longer just a point particle but shows characteristics influenced by advanced physics theories.
Advancements In Particle Physics
Particle physics has revealed that electrons are fundamental particles. They belong to a group called leptons. Electrons do not seem to have smaller parts inside. Experiments in large particle accelerators test electron properties at high energy levels. These studies confirm that electrons have a tiny mass and a negative charge. New particles related to electrons, like neutrinos, help scientists understand matter better.
Impact Of Quantum Mechanics
Quantum mechanics explains electron behavior in surprising ways. Electrons act both like particles and waves. This dual nature affects how electrons move and interact. Quantum theory introduces the idea of electron spin, a type of intrinsic angular momentum. It also describes electron clouds, where electrons are likely to be found around atoms. These concepts help explain chemical reactions and electricity flow.
Credit: quimica-tareas.fandom.com
Frequently Asked Questions
What Is An Electron Made Of?
An electron is a fundamental particle with no known substructure. It is considered elementary and not composed of smaller parts.
Does An Electron Have Internal Components?
No, electrons are elementary particles and do not have internal components or sub-particles.
What Defines An Electron’s Structure?
An electron’s structure is defined by its charge, mass, and spin, not by physical components.
Are Electrons Composed Of Quarks Or Smaller Particles?
Electrons are not made of quarks; they are distinct fundamental particles in the Standard Model.
Conclusion
Electrons are tiny particles with simple parts. They carry a negative electric charge. Inside, electrons have even smaller elements called quarks. These parts work together to give electrons their properties. Understanding electrons helps explain how atoms and matter behave. Science continues to explore these tiny building blocks.
Knowing what electrons are made of opens doors to learning more about the universe. It shows the beauty of nature at a very small scale. Electrons may be small, but they play a big role in everything around us.
Leave a Reply