When you’re diving into the fascinating world of organic chemistry, understanding how electron donating groups (EDGs) influence the highest occupied molecular orbital (HOMO) of a diene can be a game-changer. Imagine having the power to predict and control chemical reactions simply by knowing how these groups affect molecular orbitals.
Intrigued? You should be. This knowledge not only expands your understanding but also gives you an edge in designing and synthesizing new compounds. Stick around, because by the end of this article, you’ll uncover the secrets behind these chemical interactions and how they can open up new possibilities in your experiments and research.
Ready to transform your chemistry skills? Let’s dive in.
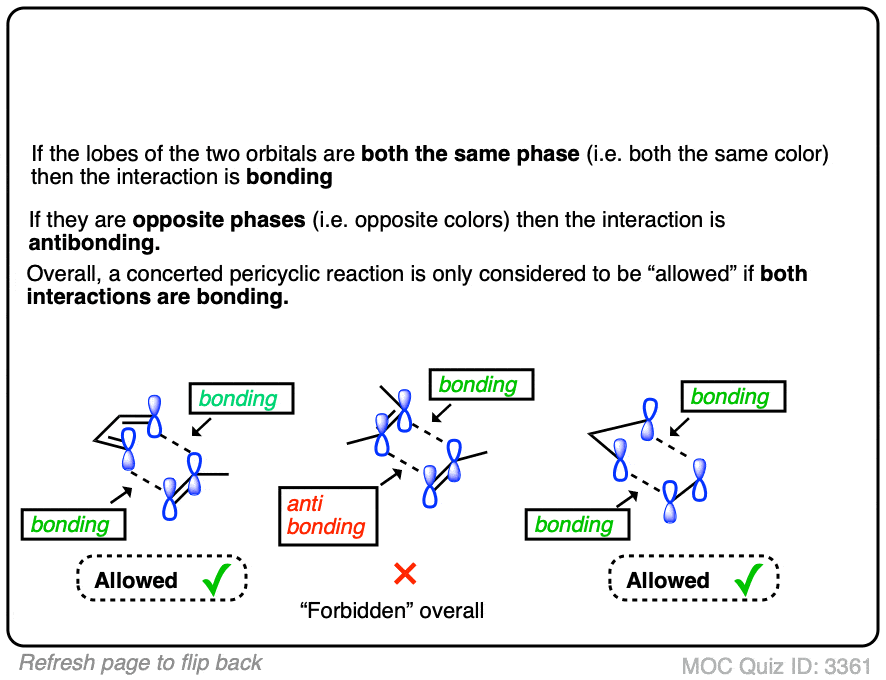
Credit: www.masterorganicchemistry.com
Electron Donating Groups In Dienes
Electron donating groups play a crucial role in modifying the electronic structure of dienes. These groups influence the Highest Occupied Molecular Orbital (HOMO) energy level. This affects how dienes behave in chemical reactions. Understanding their impact is key to controlling reaction rates and selectivity in organic synthesis.
Electron donating groups add electron density to the diene system. This changes the distribution of electrons in the molecule. The result is an increase in the HOMO energy level, making the diene more reactive in certain reactions.
Types Of Electron Donating Groups
- Alkyl Groups:Simple groups like methyl and ethyl donate electrons through sigma bonds.
- Alkoxy Groups (-OR):Oxygen atoms donate electrons through resonance and induction.
- Amino Groups (-NH2, -NR2):Nitrogen atoms donate electrons strongly through resonance.
- Hydroxyl Groups (-OH):Oxygen donates electrons via resonance and inductive effects.
- Thiol Groups (-SH):Sulfur donates electrons, similar to oxygen but less effectively.
Mechanism Of Electron Donation
Electron donating groups use resonance and induction to increase electron density.
Resonance:Lone pairs on atoms like oxygen or nitrogen overlap with the diene’s pi system. This delocalizes electrons and raises the HOMO energy.
Inductive Effect:Electron donating groups push electron density through sigma bonds. This effect is weaker but still important.
The combined effect stabilizes positive charges in transition states during reactions. This makes dienes more reactive in processes like Diels-Alder reactions.

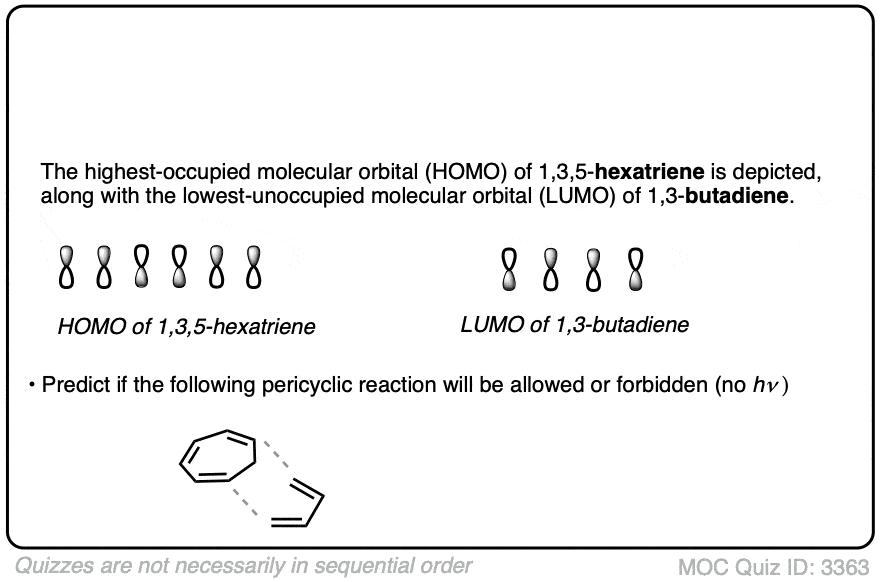
Credit: en.wikipedia.org
Homo Levels In Dienes
The Highest Occupied Molecular Orbital (HOMO) plays a key role in the chemistry of dienes. It represents the outermost electrons available for reactions. Changes in the HOMO level can affect how dienes interact with other molecules.
Electron donating groups (EDGs) attached to a diene can raise its HOMO energy. This makes the diene more reactive in many reactions. Understanding HOMO levels helps predict the behavior of dienes in different chemical environments.
Role Of Homo In Reactivity
The HOMO controls how easily a diene donates electrons. A higher HOMO means electrons are more available for bonding. This increases the diene’s reactivity in processes like Diels-Alder reactions.
Electron donating groups push electron density into the diene. This raises the HOMO energy and enhances its ability to react with electron-poor partners. The nature of the HOMO defines the diene’s chemical activity and selectivity.
Factors Influencing Homo Energy
- Type of substituents: EDGs raise HOMO levels, electron withdrawing groups lower them.
- Conjugation: Extended conjugation stabilizes and can increase HOMO energy.
- Resonance effects: Resonance donors increase electron density in the HOMO.
- Geometry: Planar dienes often have higher HOMO energy due to better overlap.
Electron donating groups interact with the diene’s pi system. This interaction shifts the HOMO energy upwards. Such shifts help explain why some dienes react faster or with different partners.
Impact Of Electron Donating Groups On Homo
Electron donating groups (EDGs) play a crucial role in altering the properties of a diene’s highest occupied molecular orbital (HOMO). These groups push electron density into the diene system, changing the energy and shape of the HOMO. Such changes affect how the diene reacts in chemical processes, especially in reactions like Diels-Alder. Understanding this impact helps predict the diene’s reactivity and selectivity.
Energy Shifts In Homo
Electron donating groups increase the energy level of the HOMO. This happens because they add electrons to the conjugated system. The raised HOMO energy makes the diene more reactive toward electron-poor partners. A higher HOMO means easier electron donation during reactions. This shift is key in controlling reaction rates and outcomes.
Effect On Electron Density
EDGs increase electron density around certain atoms in the diene. The extra electron density localizes near the donating group. This localization changes the shape and distribution of the HOMO. It enhances the nucleophilicity of the diene, making it more likely to attack electrophiles. The pattern of electron density affects where bonds form in reactions.
Reactivity Changes Due To Homo Modulation
Understanding how electron donating groups (EDGs) affect the Highest Occupied Molecular Orbital (HOMO) of a diene is key to predicting its chemical behavior. When the HOMO energy changes, the diene’s reactivity shifts, impacting how it interacts in reactions. This modulation directly influences reaction rates and outcomes, offering you a way to fine-tune your synthetic strategies.
Influence On Diels-alder Reactions
Electron donating groups raise the HOMO energy of the diene, making it more nucleophilic. This higher HOMO level allows the diene to interact more readily with electron-poor dienophiles, speeding up the Diels-Alder reaction.
Adding groups like -OH or -OCH3to your diene can boost its reactivity noticeably. You might have observed faster reaction times or higher yields when these groups are present, confirming their practical impact.
Ask yourself: could modifying the substituents on your diene optimize your reaction efficiency? Small changes here often lead to big improvements in your results.
Stability And Selectivity Alterations
Higher HOMO energy doesn’t just speed up reactions; it also changes the stability of the transition state. EDGs stabilize the developing positive charge during the Diels-Alder process, which can shift selectivity towards specific products.
This means you can influence not only how fast the reaction goes but also which product dominates. Controlling selectivity is especially important in complex syntheses where multiple products are possible.
Have you tried tweaking the electron donating character of your diene to steer the product distribution? Experimenting with different EDGs can offer a powerful handle on your reaction outcomes.
Experimental And Computational Evidence
Experimental and computational evidence provides clear insights into how electron donating groups affect the HOMO of a diene. These studies show changes in the electronic structure and reactivity. They help chemists understand the influence of substituents on molecular orbitals.
Scientists use various techniques to observe these effects. Spectroscopic methods detect changes in energy levels and electron distribution. Quantum chemical calculations model these changes and predict their impact on chemical behavior.
Spectroscopic Studies
Spectroscopic studies reveal shifts in absorption bands when electron donating groups attach to dienes. UV-Vis spectroscopy shows bathochromic shifts, indicating increased HOMO energy. This means the molecule absorbs light at longer wavelengths.
NMR spectroscopy detects changes in electron density around the diene. Electron donating groups increase electron density, altering chemical shifts. These shifts confirm the interaction between the substituent and the diene’s HOMO.
- UV-Vis spectroscopy tracks absorption wavelength changes
- NMR spectroscopy shows electron density variations
- IR spectroscopy can detect bond strength changes
Quantum Chemical Calculations
Computational methods calculate HOMO energies and visualize orbital shapes. Density Functional Theory (DFT) is commonly used for accurate predictions. Calculations show that electron donating groups raise the HOMO energy level.
These calculations compare substituted dienes with their unsubstituted forms. Results highlight increased electron density on the diene’s double bonds. This increase stabilizes the HOMO and affects reactivity in chemical reactions.
| Property | Unsubstituted Diene | Diene with Electron Donating Group |
|---|---|---|
| HOMO Energy (eV) | -6.5 | -5.8 |
| Electron Density on Double Bonds | Moderate | High |
| Reactivity | Standard | Enhanced |
Practical Applications
Electron donating groups (EDGs) influence the HOMO of dienes significantly. This effect alters the diene’s reactivity and selectivity in chemical reactions. Understanding these changes helps chemists apply dienes in various practical fields. EDGs raise the energy of the HOMO, making dienes more reactive. This property is crucial in designing molecules for specific tasks. The following sections explore how EDGs assist in designing reactive dienes and optimizing synthesis processes.
Designing Reactive Dienes
Electron donating groups increase the electron density in the diene’s HOMO. This increase enhances the diene’s ability to react with electrophiles. Chemists use this effect to create dienes that react faster and with better control.
Adding EDGs allows tuning of the diene’s reactivity to suit different reactions. For example, dienes with methoxy or amino groups often show higher reactivity. This feature helps in Diels-Alder reactions, where faster and more selective reactions are desired.
- Improved reaction rates
- Better selectivity for products
- Control over reaction pathways
These advantages make dienes with EDGs valuable in pharmaceuticals and materials science.
Synthesis Optimization
Electron donating groups help optimize the synthesis of complex molecules. By raising the HOMO energy, EDGs lower the activation energy of reactions involving dienes. This leads to higher yields and fewer side products.
EDGs allow milder reaction conditions, reducing the need for harsh chemicals or high temperatures. This improvement saves time and costs in the lab or industry.
| Benefit | Impact |
|---|---|
| Higher Yield | More product with less waste |
| Milder Conditions | Safer and cost-effective reactions |
| Improved Selectivity | Desired product favored |
These factors make EDG-modified dienes essential in efficient chemical manufacturing.
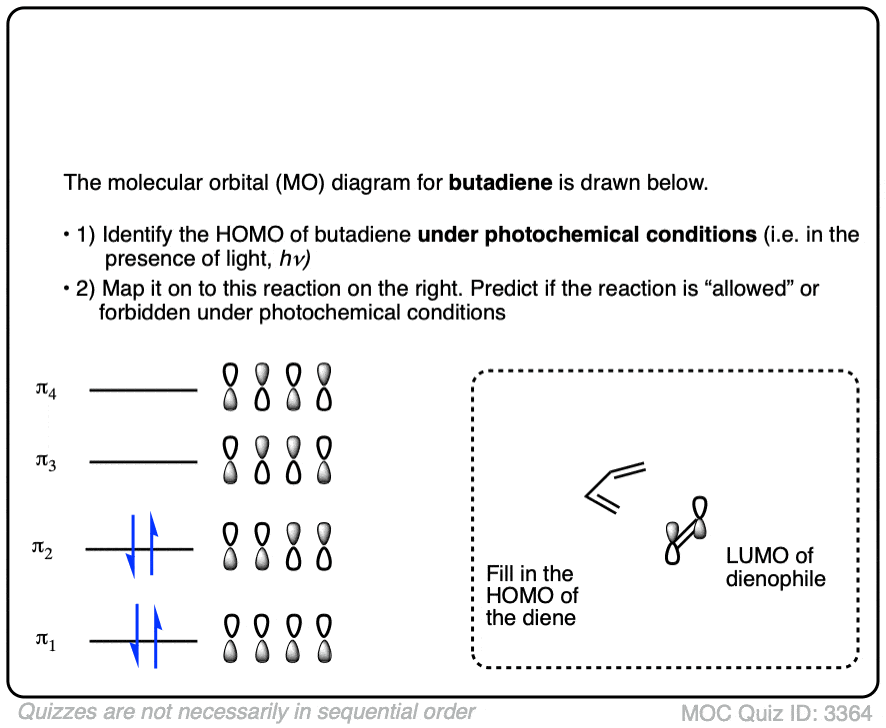
Credit: www.masterorganicchemistry.com
Frequently Asked Questions
How Do Electron Donating Groups Affect Diene Homo Energy?
Electron donating groups raise the HOMO energy of dienes by increasing electron density. This makes dienes more reactive in electrophilic reactions and enhances their stability in conjugated systems.
Why Is Homo Important In Diene Reactivity?
HOMO determines a diene’s ability to donate electrons in reactions. Higher HOMO energy leads to increased nucleophilicity, making dienes more reactive toward electrophiles.
Which Electron Donating Groups Influence Diene Homo Most?
Groups like -OH, -OCH3, and alkyl groups significantly raise diene HOMO energy. Their strong electron donation stabilizes the molecule and enhances reactivity in cycloaddition reactions.
Can Electron Donating Groups Affect Diene Stability?
Yes, electron donating groups increase diene stability by delocalizing electron density. This stabilization lowers activation energy, improving reaction rates and selectivity in synthetic applications.
Conclusion
Electron donating groups raise the HOMO energy of dienes. This makes dienes more reactive in chemical reactions. They help the molecule share electrons more easily. This effect changes how dienes interact with other chemicals. Understanding this helps in designing better chemical processes.
It also guides chemists in making new materials. Overall, electron donating groups play a key role in diene chemistry. Their influence shapes many important reactions and outcomes.
Leave a Reply