Have you ever wondered how many electrons the HOMO, or Highest Occupied Molecular Orbital, can actually hold? Understanding this can be like unlocking a secret code that explains why molecules behave the way they do.
Whether you’re a chemistry enthusiast or just curious about the tiny particles that make up the world around you, this topic is more intriguing than you might think. Imagine being able to grasp the principles that dictate molecular stability and reactivity, giving you insights into everything from the air you breathe to the food you eat.
By the end of this article, you’ll have a clear picture of how HOMO works and why it matters. Keep reading, and let’s delve into the fascinating world of electrons and orbitals together.
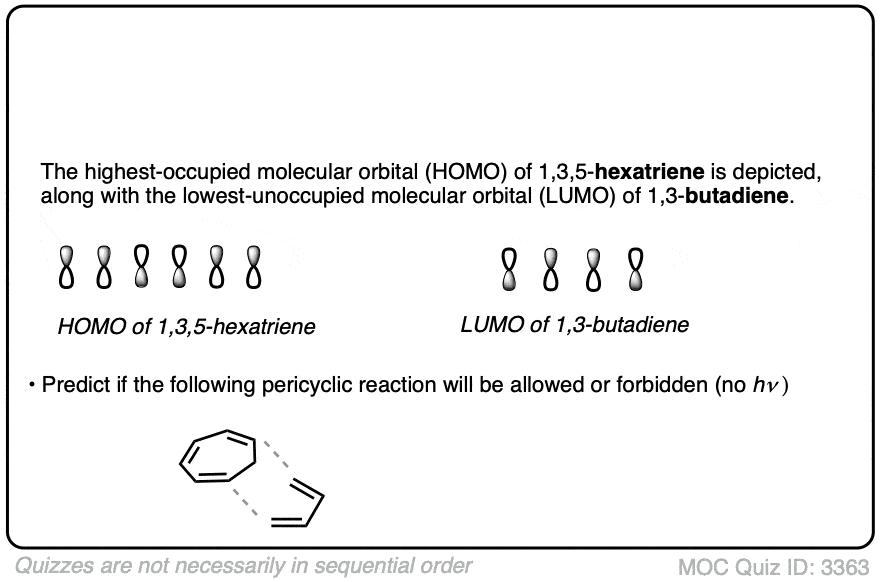
Credit: www.masterorganicchemistry.com
What Is Homo
Understanding what the Highest Occupied Molecular Orbital (HOMO) is can change how you view chemical reactions and molecular behavior. HOMO is a key concept in chemistry that helps explain how molecules interact with each other. It represents the molecular orbital that contains the highest energy electrons which are still bound to the molecule.
Think of HOMO as the outermost layer of electrons in a molecule. These electrons are the most available for chemical reactions because they require the least amount of energy to be removed or shared. Knowing about HOMO can help you predict how a molecule might react in different situations.
Definition Of Homo
The HOMO is the molecular orbital with the highest energy level that is occupied by electrons. It plays a crucial role in determining how molecules interact during chemical reactions. Electrons in the HOMO are often involved in bonding and electron transfer processes.
Why Homo Matters In Chemistry
HOMO electrons are the most reactive because they are the closest to being free. When molecules react, these electrons are usually the first to be shared or lost. This makes HOMO a critical factor in understanding reaction mechanisms and predicting outcomes.
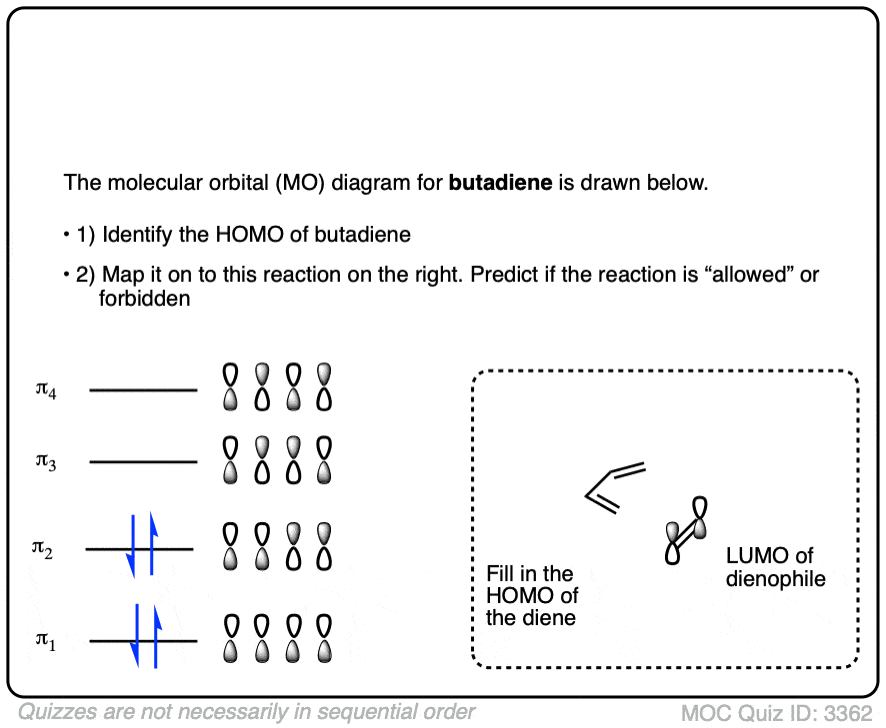
How Many Electrons Can Homo Hold?
The number of electrons that HOMO can hold depends on the specific molecule and its electronic structure. Typically, each molecular orbital, including HOMO, can hold up to two electrons with opposite spins. This simple rule helps you determine electron distribution in molecules.
Examples Of Homo In Common Molecules
- In oxygen (O2), the HOMO contains two unpaired electrons, making it highly reactive.
- In benzene, the HOMO is a delocalized orbital with six electrons, contributing to its stability.
- Water’s HOMO holds two electrons mainly from the oxygen atom, which can participate in hydrogen bonding.
Have you ever wondered why some molecules react instantly while others seem inert? Looking at their HOMO gives you the answer. Understanding HOMO not only helps you grasp basic chemistry but also guides practical applications like designing new drugs or materials.

Credit: onlinelibrary.wiley.com
Electron Capacity Basics
Understanding electron capacity is important in chemistry and biology. Electrons are tiny particles that orbit atoms in areas called orbitals. Each orbital can hold a certain number of electrons. Knowing these limits helps explain how atoms bond and interact.
The highest occupied molecular orbital (HOMO) is the outermost orbital with electrons. It plays a key role in chemical reactions. The number of electrons the HOMO can hold depends on the type of orbital involved.
What Defines Electron Capacity?
Electron capacity depends on the shape and energy of orbitals. Each orbital type has a fixed maximum number of electrons it can contain.
- s orbitals can hold up to 2 electrons
- p orbitals hold up to 6 electrons
- d orbitals hold up to 10 electrons
- f orbitals hold up to 14 electrons
Electron Capacity In Molecular Orbitals
Molecular orbitals form when atoms combine. They share electrons between atoms. The HOMO is the highest level filled with electrons in these molecular orbitals.
The maximum number of electrons in the HOMO depends on how many atomic orbitals combine. This varies by molecule structure.
Why Electron Capacity Matters
Electron capacity controls how atoms and molecules behave. It affects chemical bonding and reactivity. The HOMO’s electron count helps predict molecule reactions and stability.
Homo And Electron Configuration
The Highest Occupied Molecular Orbital (HOMO) plays a crucial role in understanding how electrons are arranged in molecules. It represents the outermost electron level that holds electrons before they jump to higher energy states or participate in chemical reactions.
Knowing the electron configuration of the HOMO helps you predict how a molecule will behave, especially in reactions involving electron transfer. The question many ask is: how many electrons can the HOMO actually hold?
What Is The Electron Capacity Of The Homo?
The HOMO, like any molecular orbital, follows a simple rule: it can hold a maximum of two electrons. These electrons must have opposite spins, complying with the Pauli Exclusion Principle.
Even if a molecule has many electrons, the HOMO itself will never hold more than two. So, when you’re analyzing a molecule, focus on these two electrons in the HOMO to understand its reactivity.
How Electron Configuration Determines Homo Energy
Electron configuration orders electrons from lower to higher energy orbitals. The HOMO is the highest energy orbital that contains electrons, making it the frontier orbital for many chemical processes.
Changes in electron configuration can shift which orbital is the HOMO. This shift affects how easily the molecule donates or accepts electrons, altering its chemical behavior.
Impact Of Electron Configuration On Chemical Properties
Your molecule’s electron configuration, especially the electrons in the HOMO, influences properties like ionization potential and bond strength. The ease with which electrons leave the HOMO can dictate how reactive a molecule is.
Understanding this can help you predict reaction pathways and design molecules with specific functions, such as better catalysts or more efficient solar cells.
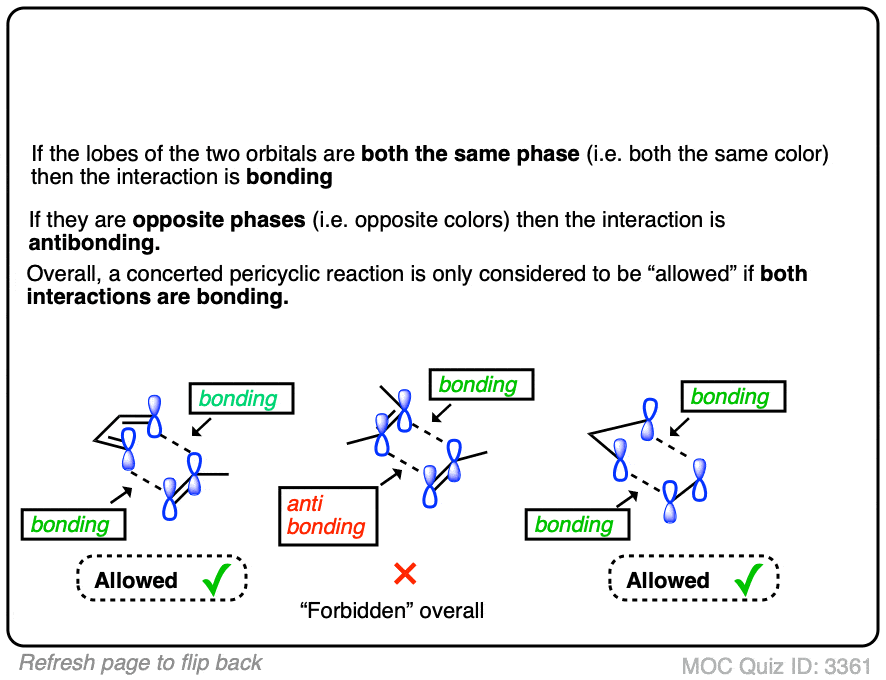
Credit: www.masterorganicchemistry.com
Factors Affecting Electron Holding
Understanding how many electrons the Highest Occupied Molecular Orbital (HOMO) can hold depends on several key factors. These factors shape the electron capacity and behavior within the orbital, influencing molecular stability and reactivity. Let’s break down these elements to see how they affect electron holding in the HOMO.
Molecular Orbital Theory
Molecular Orbital (MO) theory explains how atomic orbitals combine to form molecular orbitals, including the HOMO. The number of electrons the HOMO can hold depends on the types of atomic orbitals involved and how they overlap. Stronger overlap creates more stable orbitals that can accommodate electrons more effectively.
Think about a molecule like oxygen (O₂). Its HOMO is formed from the combination of p orbitals, which allows it to hold up to two electrons with opposite spins. This interaction dictates not just capacity but also the chemical properties of the molecule.
Energy Levels And Stability
The energy level of the HOMO impacts how many electrons it can hold comfortably. Electrons prefer to occupy lower energy, more stable orbitals first. If the HOMO is at a higher energy level, it can become less stable, limiting how many electrons it can hold without causing instability.
Have you noticed how certain molecules are more reactive? That often links back to their HOMO being higher in energy, making it easier for electrons to leave or be shared. This energy factor directly influences electron holding and molecular behavior.
Electron Spin And Pauli Principle
The Pauli Exclusion Principle states that no two electrons in the same orbital can have the same spin. This means the HOMO can hold a maximum of two electrons, each with opposite spins. This fundamental rule limits electron capacity but also ensures stability.
Imagine trying to fit more than two electrons into the HOMO—it just isn’t allowed by quantum mechanics. This spin restriction forces electrons to fill other orbitals, influencing the overall electronic structure of the molecule.
Examples In Common Molecules
Understanding how many electrons the Highest Occupied Molecular Orbital (HOMO) can hold is easier when you see it in real molecules you might know. Each molecule has a unique structure that influences its electron arrangement, especially in the HOMO. Let’s look at some common molecules to see how this works in practice and what it means for their chemical behavior.
Oxygen Molecule
The oxygen molecule (O2) has a HOMO that holds two electrons. These electrons are found in an antibonding πorbital, which plays a key role in oxygen’s magnetic properties.
If you’ve ever wondered why oxygen is paramagnetic, this electron arrangement explains it. The two unpaired electrons in the HOMO allow O2to be attracted to a magnetic field, a rare trait among molecules.
Benzene Ring
Benzene (C6H6) features a HOMO filled with six electrons. These electrons occupy a delocalized π orbital spread evenly over the ring, contributing to benzene’s stability.
This delocalization means the electrons are not stuck between two atoms but move around the ring. Have you noticed how benzene resists reactions that would disrupt this electron cloud? That’s the power of a fully occupied HOMO in aromatic systems.
Transition Metal Complexes
In transition metal complexes, the HOMO can hold varying numbers of electrons depending on the metal and its ligands. These HOMOs often involve d orbitals, which can accommodate up to 10 electrons.
Think about a complex with a metal center like iron or platinum; the electrons in the HOMO define how the complex reacts with other molecules. If you’re working with catalysts, understanding the HOMO electron count can help predict reactivity and selectivity.
Measuring Electron Capacity
Measuring the electron capacity of the Highest Occupied Molecular Orbital (HOMO) is essential to understanding a molecule’s reactivity and stability. Scientists use various techniques to determine how many electrons the HOMO can hold, which helps predict how molecules will interact in chemical reactions. You might wonder, how exactly do researchers pinpoint this electron capacity with precision? Let’s dive into the tools they rely on.
Spectroscopic Techniques
Spectroscopy is a powerful way to observe electron behavior directly. Techniques like Ultraviolet-Visible (UV-Vis) and Photoelectron Spectroscopy (PES) provide insights into the energy levels and electron distribution within molecules.
UV-Vis spectroscopy measures how electrons absorb light, revealing the energy gap between the HOMO and the Lowest Unoccupied Molecular Orbital (LUMO). This gap indirectly reflects how many electrons the HOMO can accommodate. PES, on the other hand, ejects electrons by shining light on a molecule and measures their kinetic energy, giving a direct look at the electron density in the HOMO.
Have you ever seen how a molecule’s color changes when it gains or loses electrons? That’s spectroscopy in action, showing changes in electron occupancy in real time.
Computational Methods
Computational chemistry offers a virtual microscope to explore electron capacity without physical experiments. Quantum mechanical calculations, such as Density Functional Theory (DFT), model the electron distribution in orbitals including the HOMO.
These methods simulate how electrons fill the HOMO based on the molecule’s structure and environment. You can adjust parameters and instantly see how electron capacity shifts, which is incredibly useful for designing new molecules with specific properties.
Imagine testing dozens of molecules on your computer before ever stepping into a lab—this saves time and resources. By combining computational results with experimental data, you get a fuller picture of electron capacity and behavior.
Implications For Chemical Reactivity
The number of electrons the Highest Occupied Molecular Orbital (HOMO) can hold directly influences how molecules interact during chemical reactions. This property shapes the way molecules donate or accept electrons, impacting their reactivity and the types of bonds they form. Understanding this can help you predict how a molecule might behave in various chemical environments.
Electron Density And Reactivity
The HOMO represents the highest energy electrons available for bonding. When the HOMO holds more electrons, the molecule often has a higher electron density ready to be shared or transferred. This makes the molecule more likely to act as an electron donor in reactions.
Think about how some molecules seem eager to react with acids or electrophiles. This eagerness often comes from a HOMO filled with electrons that are easy to give away. Have you noticed how molecules with high electron density tend to be more reactive in your experiments?
Impact On Electrophilic And Nucleophilic Behavior
The number of electrons in the HOMO affects whether a molecule behaves as a nucleophile or not. A HOMO rich in electrons means the molecule can easily attack positively charged or electron-poor species. This is why nucleophiles often have a filled or nearly filled HOMO.
On the other hand, if the HOMO has fewer electrons, the molecule might struggle to donate electrons, reducing its nucleophilicity. This insight can guide you when selecting reactants for a synthesis reaction or predicting reaction outcomes.
Predicting Reaction Pathways
Knowing how many electrons the HOMO can hold helps you anticipate which reaction pathways are favorable. Molecules with a HOMO capable of holding more electrons can engage in reactions that involve electron donation, such as substitution or addition reactions.
This knowledge allows you to tailor reaction conditions or choose catalysts that favor the desired pathway. Have you ever adjusted a reaction based on how reactive you expected the molecule to be? Understanding HOMO electron capacity sharpens this intuition.
Influence On Molecular Stability
A filled HOMO can sometimes lead to increased reactivity but decreased stability. Molecules with a HOMO near full capacity may be more reactive but less stable under certain conditions. This balance between stability and reactivity is critical in designing molecules for practical applications.
When working with reactive intermediates, consider how the electron count in the HOMO affects their lifespan. This can help you control reaction rates and yields more effectively.
Frequently Asked Questions
How Many Electrons Can The Homo Hold?
The Highest Occupied Molecular Orbital (HOMO) can hold up to two electrons. This follows the Pauli exclusion principle, allowing two electrons with opposite spins in one orbital.
Why Is Electron Count In Homo Important?
Electron count in HOMO influences molecular reactivity. It determines how molecules interact in chemical reactions and affects electronic properties.
Can Homo Hold More Than Two Electrons?
No, the HOMO cannot hold more than two electrons. Each molecular orbital, including HOMO, accommodates a maximum of two electrons with opposite spins.
How Does Homo Affect Molecular Stability?
HOMO electrons are crucial for stability. Molecules with fully paired electrons in HOMO tend to be more stable and less reactive.
Conclusion
The HOMO can hold a limited number of electrons, which affects molecule behavior. Understanding this helps explain chemical reactions and stability. Electrons in the HOMO are key players in bonding and energy transfer. Knowing their capacity gives insight into how molecules interact.
This knowledge supports deeper learning in chemistry and materials science. Keep exploring to see how electron capacity influences different molecules. Simple steps reveal complex chemical concepts.
Leave a Reply