Have you ever wondered how your body turns food into energy? At the heart of this process lies the electron transport chain, a vital step in producing the energy you need to live and thrive.
But what exactly are the starting components of this chain, and why do they matter so much? Understanding these key players can give you a clearer picture of how energy flows inside your cells. Keep reading, and you’ll uncover the building blocks that set this amazing process in motion—knowledge that can deepen your appreciation for the science powering your everyday life.
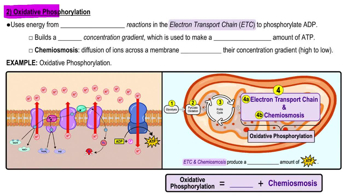
Credit: www.pearson.com
Electron Transport Chain Basics
The electron transport chain (ETC) is a crucial part of cellular respiration. It helps produce energy in the form of ATP. Found in the mitochondria, it is essential for life. The ETC transfers electrons through a series of complexes. Each complex plays a unique role in energy production.
Complex I: Nadh Dehydrogenase
Complex I is the starting point of the electron transport chain. It accepts electrons from NADH. These electrons come from earlier stages of cellular respiration. Complex I then transfers these electrons to the next complex. This process helps in pumping protons across the mitochondrial membrane.
Complex Ii: Succinate Dehydrogenase
Complex II also participates in the electron transport chain. It receives electrons from FADH2. These electrons are generated during the Krebs cycle. Unlike Complex I, it does not pump protons. Still, it plays a vital role in transferring electrons to the next complex.
Coenzyme Q: Ubiquinone
Coenzyme Q acts as an electron shuttle. It transfers electrons between Complex I and III. It also moves electrons from Complex II to III. Coenzyme Q is vital for maintaining the electron flow.
Complex Iii: Cytochrome Bc1
Complex III plays a central role in the chain. It accepts electrons from Coenzyme Q. Then, it transfers them to Cytochrome c. This complex also pumps protons, helping create a gradient.
Cytochrome C
Cytochrome c is a small protein. It carries electrons from Complex III to Complex IV. It plays a critical role in ensuring electron flow continuity. Without it, the chain would halt.
Complex Iv: Cytochrome C Oxidase
Complex IV is the final stop for electrons. It transfers them to oxygen, forming water. This step completes the electron transport chain process. It also pumps protons, contributing to ATP synthesis.

Credit: www.dreamstime.com
Role Of Nadh Dehydrogenase
The role of NADH dehydrogenase is crucial as the starting point of the electron transport chain. It acts as the first enzyme complex that accepts electrons from NADH, initiating the chain of reactions that ultimately produce energy. Understanding how this enzyme functions helps you grasp how your cells generate the power they need to survive.
Structure And Function
NADH dehydrogenase, also known as Complex I, is a large protein complex embedded in the inner mitochondrial membrane. It has multiple subunits that work together to carry out its function efficiently.
This complex accepts electrons from NADH, which comes from metabolic processes like glycolysis and the Krebs cycle. It then transfers these electrons to a small molecule called ubiquinone, while simultaneously pumping protons across the membrane. This proton pumping creates a gradient that drives ATP production, the cell’s energy currency.
Electron Transfer Process
The electron transfer process starts when NADH binds to NADH dehydrogenase and donates two electrons. These electrons move through a series of iron-sulfur clusters within the complex, which act like stepping stones.
As the electrons pass through these clusters, energy is released and used to pump protons from the mitochondrial matrix to the intermembrane space. This movement is essential for building the electrochemical gradient your cells need to produce ATP.
Have you ever wondered how a simple transfer of electrons can power your entire body? The efficiency of NADH dehydrogenase in managing this transfer is a key part of that process.
Function Of Succinate Dehydrogenase
Succinate dehydrogenase plays a key role in the electron transport chain. It acts as a bridge between the Krebs cycle and the chain. This enzyme helps in transferring electrons from succinate to the electron transport system. Its function ensures smooth energy flow in cells.
Integration With Krebs Cycle
Succinate dehydrogenase is part of the Krebs cycle. It catalyzes the conversion of succinate to fumarate. This step produces electrons by removing hydrogen atoms. Unlike other enzymes, it is embedded in the inner mitochondrial membrane. This placement allows it to link directly to the electron transport chain. It connects the metabolic cycle to energy production in mitochondria.
Electron Donation Mechanism
The enzyme passes electrons to flavin adenine dinucleotide (FAD). FAD accepts these electrons and becomes FADH2. Then, FADH2 transfers electrons to coenzyme Q (ubiquinone). Coenzyme Q carries electrons further into the chain. This process helps create a proton gradient. The gradient powers ATP synthesis, the main energy currency of cells.
Ubiquinone As An Electron Carrier
Ubiquinone plays a vital role as an electron carrier in the electron transport chain. It acts as a mobile molecule, shuttling electrons between protein complexes. This movement helps produce energy efficiently in cells. Understanding ubiquinone’s properties and how it moves clarifies its importance in cellular respiration.
Chemical Properties
Ubiquinone is a small, lipid-soluble molecule. It has a long tail made of isoprene units. This tail helps it stay within the inner mitochondrial membrane. The molecule contains a quinone group that can accept and release electrons.
It exists in three forms:
- Oxidized form (ubiquinone)
- Partially reduced form (semiquinone)
- Fully reduced form (ubiquinol)
This ability to switch forms allows ubiquinone to carry electrons effectively. It also transfers protons across the membrane, contributing to the proton gradient.
Movement Between Complexes
Ubiquinone moves freely within the inner mitochondrial membrane. It carries electrons from Complex I and Complex II to Complex III. This mobility is crucial for continuous electron flow in the chain.
Its lipid-soluble nature allows it to diffuse smoothly through the membrane. This ensures quick and efficient electron transfer. Ubiquinone’s movement supports the production of ATP, the cell’s energy currency.
Initial Electron Acceptors
The electron transport chain begins with molecules that first accept electrons. These initial electron acceptors start the process of energy conversion. They play a vital role in capturing electrons from nutrients. Without them, the chain cannot function properly.
Coenzymes And Prosthetic Groups
Coenzymes and prosthetic groups act as the first electron carriers. They are tightly bound to proteins in the chain. Coenzymes like NAD+ and FAD accept electrons from food molecules. These coenzymes become reduced by gaining electrons.
Prosthetic groups such as flavin mononucleotide (FMN) and iron-sulfur clusters also accept electrons. They help transfer electrons within protein complexes. Their structure allows easy electron movement. These groups ensure electrons flow smoothly through the chain.
Redox Reactions At Start
Redox reactions drive the electron transfer process. Oxidation means losing electrons. Reduction means gaining electrons. Initial acceptors undergo reduction by gaining electrons from coenzymes.
This electron transfer releases energy. The energy helps pump protons across membranes. This proton movement creates a gradient. The gradient powers ATP synthesis, the cell’s energy source. Thus, early redox reactions set the stage for energy production.

Credit: www.dreamstime.com
Frequently Asked Questions
What Are The First Components Of The Electron Transport Chain?
The starting components of the electron transport chain are Complex I (NADH dehydrogenase) and Complex II (succinate dehydrogenase). They receive electrons from NADH and FADH2, initiating the transfer process that drives ATP production in mitochondria.
How Does Complex I Start The Electron Transport Chain?
Complex I accepts electrons from NADH and transfers them to ubiquinone. This process pumps protons across the mitochondrial membrane, creating a gradient essential for ATP synthesis.
What Role Does Complex Ii Play In Starting The Chain?
Complex II receives electrons from FADH2 and passes them to ubiquinone. Unlike Complex I, it does not pump protons but contributes electrons to the chain for energy production.
Why Is Ubiquinone Important In The Electron Transport Chain?
Ubiquinone acts as a mobile electron carrier between Complexes I and II to Complex III. It helps transfer electrons while maintaining the proton gradient needed for ATP generation.
Conclusion
The electron transport chain starts with key components that move electrons. These parts include complex I and complex II. They pass electrons to coenzyme Q, which then sends them further. This chain helps create energy cells need to work. Understanding these basics makes the whole process clearer.
It shows how cells turn food into usable power. Simple steps lead to big results inside your body. The electron transport chain plays a vital role in life.
Leave a Reply