Have you ever wondered exactly where oxygen is used in the electron transport chain? If you’re curious about how your cells turn food into energy, understanding oxygen’s role is key.
This process happens deep inside your cells and is essential for keeping you alive and full of energy. By the end of this article, you’ll clearly know where oxygen comes into play, why it matters, and how it helps your body produce the energy you need every second.
Ready to uncover this vital step in cellular energy production? Let’s dive in!
Credit: bioprinciples.biosci.gatech.edu
Role Of Oxygen In Cellular Respiration
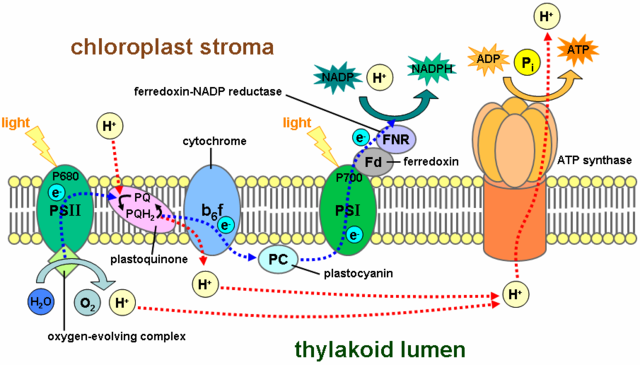
Oxygen plays a crucial role in cellular respiration, especially within the electron transport chain (ETC). Without oxygen, the entire process of producing energy in your cells would come to a halt. Understanding exactly where and how oxygen is consumed helps you appreciate how your body efficiently extracts energy from the food you eat.
In the electron transport chain, oxygen acts as the final electron acceptor. Electrons travel through a series of protein complexes embedded in the mitochondrial membrane. At the end of this chain, oxygen accepts these electrons, allowing the chain to continue moving.
Without oxygen to accept electrons, the ETC would become backed up, and energy production would stop. This is why oxygen is essential for aerobic respiration and why your body demands it constantly.
Formation Of Water In The Electron Transport Chain
When oxygen accepts electrons, it combines with protons (hydrogen ions) to form water. This reaction happens at Complex IV, also known as cytochrome c oxidase. The formation of water is a critical step that keeps the electron transport chain running smoothly.
Think about the last time you held your breath—your body quickly signals you to breathe in again because oxygen is needed to keep producing energy. This water formation prevents a dangerous buildup of electrons and maintains the cell’s energy flow.
Impact On Atp Production
The role of oxygen directly influences how much ATP, the energy currency of cells, is produced. Oxygen’s acceptance of electrons allows proton pumps in the ETC to create a proton gradient. This gradient drives ATP synthase to produce ATP efficiently.
If oxygen is absent, the proton gradient collapses, and ATP production drops dramatically. This is why your muscles fatigue during intense exercise when oxygen supply can’t meet demand.
Credit: www.pearson.com
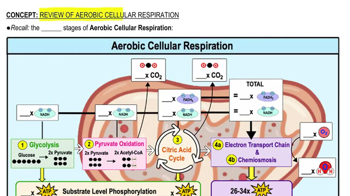
Electron Transport Chain Basics
The electron transport chain (ETC) is a crucial process in cell respiration. It takes place in the inner membrane of mitochondria. The ETC helps produce energy by transferring electrons through protein complexes. This transfer creates a flow that powers the production of ATP, the cell’s energy currency.
Oxygen plays a key role in this chain. It acts as the final electron acceptor. Without oxygen, the electron flow would stop, and energy production would fail. Understanding the basics of the ETC helps clarify where oxygen is consumed.
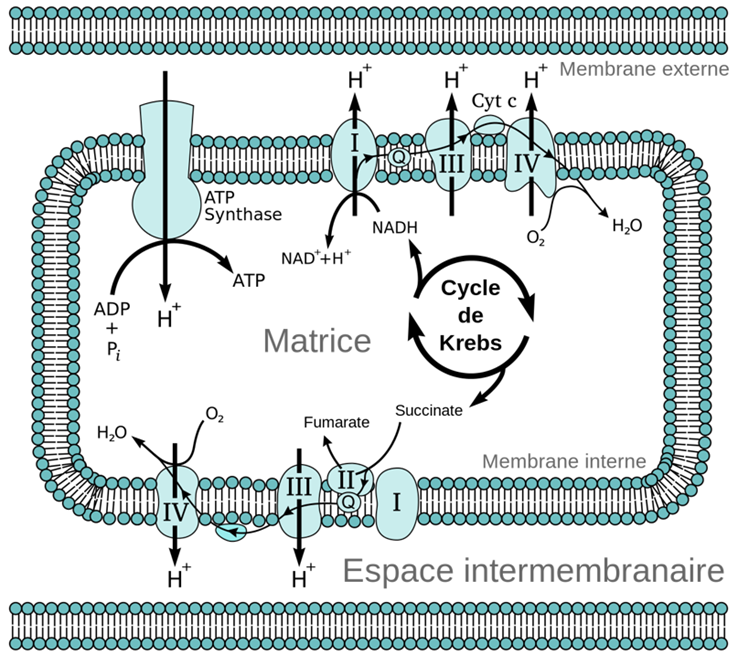
Structure Of The Electron Transport Chain
The ETC consists of four main protein complexes (I to IV) and two mobile carriers. These proteins work together to move electrons from NADH and FADH2 to oxygen. The chain is embedded in the mitochondrial inner membrane.
- Complex I:Receives electrons from NADH.
- Complex II:Receives electrons from FADH2.
- Complex III:Transfers electrons from carriers to Complex IV.
- Complex IV:Transfers electrons to oxygen.
Role Of Oxygen In The Electron Transport Chain
Oxygen is essential for the ETC to function. It accepts electrons at Complex IV. This acceptance allows the chain to continue moving electrons. Oxygen combines with electrons and protons to form water. This step is vital to keep the electron flow going.
Without oxygen, electrons would back up. This stops ATP production and energy supply. The presence of oxygen ensures efficient energy generation in cells.
Energy Production In The Electron Transport Chain
As electrons move through the ETC, energy is released. This energy pumps protons from the mitochondrial matrix to the intermembrane space. The proton gradient created powers ATP synthase, an enzyme that makes ATP.
ATP synthase uses the flow of protons back into the matrix. This flow drives the conversion of ADP to ATP. The whole process depends on oxygen to maintain the electron flow and proton gradient.
Oxygen As The Final Electron Acceptor
Oxygen plays a crucial role in the electron transport chain (ETC) as the final electron acceptor. Without oxygen, the entire chain would back up, stopping the production of energy your cells need to function. Understanding where and how oxygen consumes electrons can help you appreciate the fine-tuned process that keeps you alive every second.
What Happens To Oxygen In The Electron Transport Chain?
At the end of the ETC, electrons have traveled through a series of protein complexes. These electrons need a place to go; otherwise, the chain stalls. Oxygen accepts these electrons and combines with protons (H+) to form water.
This reaction is essential because it removes low-energy electrons from the system, allowing the chain to continue flowing smoothly. Without oxygen, electrons would accumulate, and energy production would cease.
Why Is Oxygen Called The Final Electron Acceptor?
Oxygen’s role as the final electron acceptor means it is the last molecule to receive electrons in the ETC. Think of it as the finishing point in a relay race where the baton (electrons) is passed along.
Since oxygen has a high affinity for electrons, it easily pulls them through the chain. This pull creates a flow that powers the pumping of protons across the membrane, a key step for ATP synthesis.
How Does Oxygen Consumption Affect Your Energy Levels?
Every time you breathe in, oxygen enters your bloodstream and reaches your cells. There, it accepts electrons in the ETC, enabling your mitochondria to produce ATP—the energy currency of your body.
If oxygen supply drops, your cells struggle to produce enough ATP, leading to fatigue and decreased performance. This is why you feel breathless during intense exercise; your muscles demand more oxygen to keep energy production high.
What Happens If Oxygen Is Not Available?
- Electrons cannot be passed to oxygen, causing a backup in the chain.
- The proton gradient needed for ATP production collapses.
- Cells switch to less efficient energy pathways, like fermentation.
This switch is not sustainable and can lead to cell damage or death if oxygen deprivation continues. Understanding this can explain why oxygen is essential, not just for survival but for optimal cellular function.
Mechanism Of Oxygen Consumption
The mechanism of oxygen consumption in the electron transport chain (ETC) is a crucial step in cellular respiration. Oxygen acts as the final electron acceptor, ensuring the entire chain functions smoothly. Understanding how oxygen is consumed reveals why it’s indispensable for energy production in your cells.
Electron Flow To Oxygen
Electrons travel through a series of protein complexes embedded in the mitochondrial membrane. These complexes pass electrons down the chain, releasing energy to pump protons across the membrane. Eventually, electrons reach Complex IV, also called cytochrome c oxidase.
At Complex IV, electrons combine with oxygen molecules. This step is critical because oxygen’s high affinity for electrons pulls them through the chain efficiently. Without oxygen, the flow of electrons would back up, halting energy production.
Formation Of Water
Once oxygen accepts electrons, it doesn’t just float away—it immediately reacts with protons (H+) to form water. This reaction prevents free oxygen radicals from damaging the cell. It also maintains the proton gradient essential for ATP synthesis.
Consider this: if oxygen didn’t form water, harmful reactive oxygen species could accumulate, causing cellular damage. This precise chemical step protects your cells while powering them. Have you ever wondered how something as simple as water formation is key to your energy and survival?
Location Of Oxygen Utilization
Oxygen plays a vital role in the electron transport chain. It acts as the final electron acceptor. Without oxygen, the chain cannot function properly. The site where oxygen is used is very specific inside the cell. Understanding this location helps explain how energy is produced efficiently.
Inner Mitochondrial Membrane
The electron transport chain is located on the inner mitochondrial membrane. This membrane is folded into structures called cristae. These folds increase the surface area, allowing more space for the chain’s components. Oxygen is consumed right here in this membrane during the last step of the chain.
Electrons move along protein complexes embedded in this membrane. As electrons pass through, they release energy. This energy pumps protons across the membrane, creating a gradient. Oxygen accepts electrons at the end of this chain, enabling the process to continue.
Cytochrome C Oxidase Complex
Oxygen utilization happens specifically at the cytochrome c oxidase complex. This is the last enzyme in the electron transport chain. It receives electrons from cytochrome c molecules. The complex then transfers these electrons to oxygen molecules.
Oxygen combines with electrons and protons to form water. This reaction is crucial for maintaining the flow of electrons. Without cytochrome c oxidase, oxygen could not be used effectively. The enzyme ensures oxygen acts as a strong electron acceptor, supporting continuous energy production.
Importance Of Oxygen In Atp Production
Oxygen plays a vital role in the production of ATP in cells. ATP is the main energy source for many cellular functions. Without oxygen, cells cannot produce enough ATP to meet their energy needs.
In the electron transport chain, oxygen acts as the final electron acceptor. This step is crucial for the chain to continue working. Oxygen combines with electrons and protons to form water, a harmless byproduct.
This process helps maintain the flow of electrons through the chain. Without oxygen, the electron transport chain would stop, and ATP production would drop drastically.
How Oxygen Accepts Electrons In The Chain
At the end of the electron transport chain, oxygen receives electrons. It binds with these electrons and hydrogen ions. This reaction creates water, which leaves the cell safely.
This electron acceptance keeps the chain moving. It allows proteins in the chain to pump protons. These protons create a gradient used to make ATP.
The Role Of Oxygen In Maintaining Proton Gradient
The proton gradient powers ATP synthase, the enzyme that makes ATP. Oxygen helps maintain this gradient by removing electrons. Without oxygen, protons would build up and stop the chain.
Oxygen ensures a steady flow of protons across the membrane. This flow drives the production of ATP, providing energy for the cell.
Consequences Of Oxygen Deficiency On Atp Production
When oxygen is low, the electron transport chain slows down. ATP production decreases, and cells struggle to get energy. This condition is known as hypoxia.
Cells then rely on less efficient processes, like fermentation. These processes produce much less ATP and can lead to fatigue and damage.
Consequences Of Oxygen Deficiency
Oxygen plays a vital role in the electron transport chain by accepting electrons at the end of the process. Without enough oxygen, cells cannot complete this chain efficiently. This lack of oxygen leads to several serious effects on cellular functions and energy production.
Impact On Electron Transport Chain
Oxygen shortage stops the electron transport chain from working properly. Electrons cannot be passed to oxygen, causing a backlog in the chain. This blockage reduces the ability to create ATP, the cell’s energy currency. The chain becomes less efficient and may even generate harmful reactive oxygen species. Cells struggle to maintain energy levels without oxygen at the final step.
Effects On Cellular Metabolism
Low oxygen levels force cells to switch to less efficient energy methods. They rely more on anaerobic respiration, which produces less ATP. This process also creates lactic acid, leading to muscle fatigue and soreness. Cells may suffer from energy shortages, affecting growth and repair. Long-term oxygen deficiency can damage tissues and impair organ function.

Credit: www.researchgate.net
Frequently Asked Questions
Where Is Oxygen Consumed In The Electron Transport Chain?
Oxygen is consumed at Complex IV (cytochrome c oxidase) in the electron transport chain. It acts as the final electron acceptor, combining with electrons and protons to form water, which is essential for maintaining the chain’s function and ATP production.
Why Does Oxygen Act As The Final Electron Acceptor?
Oxygen has a high affinity for electrons, allowing it to accept electrons after they pass through Complex IV. This acceptance prevents electron backup and enables continuous ATP synthesis by maintaining the flow of electrons through the chain.
What Happens If Oxygen Is Not Available In The Electron Transport Chain?
Without oxygen, the electron transport chain stops functioning. Electrons cannot be transferred to the final acceptor, causing ATP production to halt and leading to cellular energy deficiency and potential cell death.
How Does Oxygen Consumption Affect Atp Production?
Oxygen consumption drives the electron transport chain, creating a proton gradient that powers ATP synthase. Without oxygen, this gradient collapses, stopping ATP synthesis and reducing cellular energy output significantly.
Conclusion
Understanding oxygen’s role in the electron transport chain is crucial. It acts as the final electron acceptor. This process is vital for energy production in cells. Without oxygen, the chain would halt. Energy production would drop significantly. This knowledge helps in biology and medicine fields.
It aids in grasping how our bodies produce energy. By knowing these basics, one appreciates cellular respiration better. It’s fascinating how oxygen supports life at the cellular level. This insight enhances our understanding of essential biological processes. Keep exploring these wonders of science.
Leave a Reply