Which component in the electron transport chain is the key to powering your cells? If you’ve ever wondered how your body turns food into energy, understanding this process is essential.
The electron transport chain might sound complex, but breaking down its components will give you a clearer picture of how life’s energy flows. By the end of this article, you’ll know exactly which part plays the crucial role in energy production—and why it matters to your health and vitality.
Keep reading to unlock the secrets behind your body’s powerhouse.
Credit: www.droracle.ai
Electron Transport Chain Overview
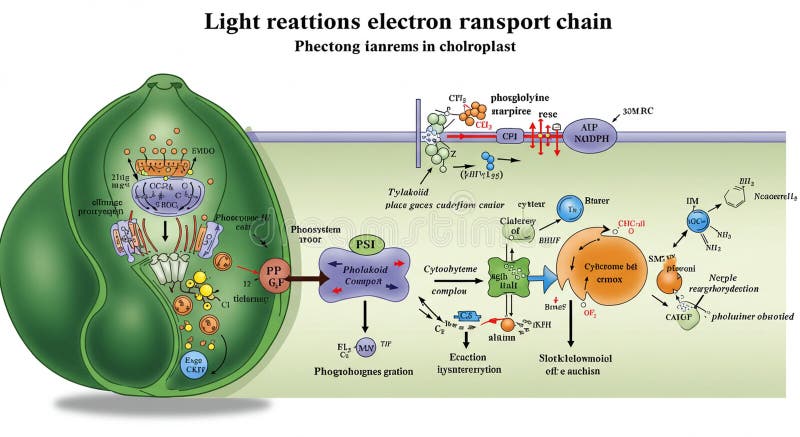
The electron transport chain (ETC) is a vital process in cells. It helps produce energy by moving electrons through a series of proteins. This chain is located in the inner membrane of mitochondria, the cell’s powerhouses.
During the ETC, electrons pass from one protein complex to another. This movement creates a flow that helps pump protons across the membrane. The result is a proton gradient that drives the production of ATP, the main energy molecule in cells.
Understanding the components of the ETC reveals how cells convert food into usable energy. Each part plays a specific role in making sure the process works efficiently and keeps the cell alive.
Structure And Location Of The Electron Transport Chain
The ETC is embedded in the inner membrane of mitochondria. This membrane has folds called cristae, increasing the surface area. The chain consists of four main protein complexes and two mobile carriers. These components work together to transfer electrons step-by-step.
Function Of Electron Carriers
Electrons come from molecules like NADH and FADH2. These carriers deliver electrons to the first complex in the chain. The electrons move through the complexes, losing energy gradually. This energy pumps protons into the space between membranes.
Role Of Proton Gradient In Atp Production
The proton pumping creates a high concentration of protons outside the inner membrane. Protons then flow back through ATP synthase, a protein that makes ATP. This flow provides the energy to join ADP and phosphate into ATP.
Key Components Of The Chain
The electron transport chain is a series of protein complexes. These complexes work together to create energy in cells. Each component plays a specific role in moving electrons and pumping protons.
Understanding the key parts helps explain how cells produce ATP, the main energy source. The chain includes four main complexes and the ATP synthase enzyme. Each has unique functions but all contribute to energy creation.
Complex I
Complex I is the first protein complex in the chain. It receives electrons from NADH molecules. This complex uses energy from electrons to pump protons across the membrane. It helps create a proton gradient for ATP production.
Complex Ii
Complex II receives electrons from FADH2 molecules. It transfers these electrons to the chain without pumping protons. Complex II connects the Krebs cycle to the electron transport chain. It helps keep the electron flow continuous.
Complex Iii
Complex III transfers electrons from ubiquinol to cytochrome c. It also pumps protons to increase the proton gradient. This step is important for maintaining energy flow in the chain. Complex III acts as a bridge between complexes.
Complex Iv
Complex IV is the last protein complex in the chain. It transfers electrons to oxygen molecules. Oxygen combines with electrons and protons to form water. Complex IV also pumps protons, completing the gradient for ATP synthesis.
Atp Synthase
ATP synthase uses the proton gradient to make ATP. Protons flow back through this enzyme, driving ATP production. It converts ADP and phosphate into ATP molecules. ATP synthase is essential for cellular energy supply.
Role Of Complex I
Complex I plays a crucial role in the electron transport chain, acting as the first major site where electrons enter the system. It is responsible for initiating the chain’s energy conversion process by interacting with NADH, a key molecule in cellular respiration. Understanding Complex I’s role helps you grasp how your cells generate the energy you need every second.
Nadh Oxidation
Complex I starts by accepting electrons from NADH, which is produced during earlier stages of metabolism. It oxidizes NADH, meaning it removes electrons from it, turning NADH back into NAD+. This step is essential because it feeds electrons into the electron transport chain, allowing the energy extraction process to continue.
Without this electron transfer, the chain would stall, and your cells wouldn’t be able to produce ATP efficiently. Have you ever wondered how your muscles keep working during intense exercise? Complex I’s ability to process NADH is part of the answer.
Proton Pumping Function
While transferring electrons, Complex I also pumps protons (H+ ions) from the mitochondrial matrix to the intermembrane space. This action creates a proton gradient, which stores potential energy like water behind a dam. The energy in this gradient is later used to produce ATP, the cell’s energy currency.
The proton pumping by Complex I is vital because it sets up the conditions for ATP synthase, another protein complex, to do its job. If Complex I fails to pump protons effectively, your cells would struggle to meet their energy demands. Can you imagine what happens when this process is impaired in diseases like Parkinson’s?
Importance Of Complex Ii
Complex II plays a unique and crucial role in the electron transport chain. Unlike other complexes, it directly links the Krebs cycle to the electron transport process, making it a vital bridge in cellular energy production. Understanding its function can deepen your appreciation of how cells efficiently generate energy.
Succinate Dehydrogenase
Succinate dehydrogenase is the enzyme that forms the core of Complex II. It catalyzes the oxidation of succinate to fumarate in the Krebs cycle. This reaction is special because it transfers electrons directly to the electron transport chain.
What makes this enzyme fascinating is that it is embedded in the inner mitochondrial membrane, unlike other Krebs cycle enzymes floating freely in the matrix. This positioning allows it to hand off electrons efficiently to the chain without losing energy.
Fadh2 Contribution
Complex II is the only complex that accepts electrons from FADH2. This molecule carries fewer electrons than NADH, so it contributes less energy to the chain. However, its role is still essential because it feeds electrons into the system without pumping protons, unlike other complexes.
This means FADH2’s electrons help maintain the flow of electrons and support ATP production, but with a slightly lower yield. Have you ever wondered why different molecules produce different amounts of ATP? Complex II’s interaction with FADH2 explains part of this variation.
Significance Of Complex Iii
Complex III plays a crucial role in the electron transport chain, acting as a key hub where electron transfer helps generate the energy your cells need. Its function goes beyond simple electron movement; it directly impacts how efficiently your body produces ATP, the energy currency of cells. Understanding this complex can shed light on how energy production is finely tuned and what happens when this process is disrupted.
Electron Transfer
At Complex III, electrons are transferred from coenzyme Q (ubiquinol) to cytochrome c. This step is vital because it moves electrons closer to the final step of the chain, where oxygen is reduced to water. If electron transfer here slows down, your cells produce less ATP, which can affect everything from muscle performance to brain function.
Have you ever wondered why even small changes in your energy levels can feel so draining? Complex III’s efficiency might be the answer. It’s not just about moving electrons; it’s about maintaining a steady flow that keeps your energy steady.
Q-cycle Mechanism
The Q-cycle is a fascinating process within Complex III that maximizes the energy extracted from electrons. It involves a clever sequence where electrons from one molecule of ubiquinol are split and passed along two different pathways. This mechanism helps pump protons across the mitochondrial membrane, creating the proton gradient needed for ATP synthesis.
Think of the Q-cycle as a smart energy-saving trick your cells use. Without it, the cell would waste potential energy, leading to less ATP and more fatigue. Understanding the Q-cycle can help you appreciate how even tiny molecular details impact your overall energy and health.

Credit: library.fiveable.me
Functionality Of Complex Iv
Complex IV plays a vital role in the electron transport chain. It helps convert energy stored in electrons into a usable form for cells. This complex is the last step in the chain, where electrons combine with oxygen to form water. Its activity is crucial for maintaining the energy flow needed for cellular functions.
Cytochrome C Oxidase
Cytochrome c oxidase is the main enzyme in Complex IV. It accepts electrons from cytochrome c molecules. These electrons then move through the enzyme’s metal centers. This process drives the pumping of protons across the membrane. The proton movement creates a gradient used to produce ATP, the cell’s energy currency.
Oxygen Reduction
Oxygen acts as the final electron acceptor in Complex IV. The enzyme facilitates the reduction of oxygen to water. This step prevents the buildup of harmful free radicals. It also ensures the continuation of electron flow. Without oxygen reduction, energy production would stop, leading to cell damage.
Critical Role Of Atp Synthase
ATP synthase plays a vital role in the electron transport chain, acting as the key enzyme that produces ATP, the energy currency of your cells. Without ATP synthase, the energy generated by electron transport would never be captured effectively. Understanding its function can change how you view cellular energy and its efficiency.
Chemiosmotic Theory
The chemiosmotic theory explains how ATP synthase uses a proton gradient to make ATP. As electrons move through the electron transport chain, protons are pumped across the mitochondrial membrane, creating a difference in proton concentration.
This proton gradient stores potential energy, which ATP synthase harnesses by allowing protons to flow back into the mitochondrial matrix. This flow drives the enzyme to synthesize ATP from ADP and inorganic phosphate.
Have you ever considered how this tiny molecular machine converts a simple flow of protons into usable energy? This process highlights the elegance of biological energy conversion.
Atp Production
ATP synthase produces ATP by rotating parts of its structure as protons pass through it. This mechanical rotation facilitates chemical reactions that bind ADP with phosphate, forming ATP.
The amount of ATP produced depends on the number of protons flowing through ATP synthase. Typically, one full rotation synthesizes about three ATP molecules.
- ATP production is tightly linked to oxygen availability since oxygen drives the electron transport chain.
- Without oxygen, the proton gradient collapses, and ATP synthase cannot function efficiently.
- This explains why your cells require oxygen for sustained energy production.
Next time you feel tired after intense activity, remember that your ATP synthase might be running low on fuel to keep your cells energized.
Comparative Analysis Of Components
The Electron Transport Chain (ETC) contains several key components. Each plays a distinct role in energy production within cells. Comparing these components helps understand their unique functions and contributions. This analysis covers their energy yield, efficiency, and regulation.
Energy Yield
Each component in the ETC transfers electrons and helps create ATP. Complex I and Complex II start the process by passing electrons to Coenzyme Q.
- Complex I:Pumps protons and contributes to high ATP production.
- Complex II:Does not pump protons but feeds electrons into the chain.
- Complex III:Pumps protons and transfers electrons to Cytochrome c.
- Complex IV:Pumps protons and transfers electrons to oxygen, forming water.
Complex I, III, and IV generate most of the proton gradient. This gradient powers ATP synthase to make ATP. Complex II contributes less to ATP yield.
Efficiency And Regulation
The ETC components vary in efficiency and control mechanisms. Efficiency depends on electron transfer speed and proton pumping ability.
- Complex I:Efficient but sensitive to damage and inhibition.
- Complex II:More stable but less efficient in proton pumping.
- Complex III:Efficient and critical for electron transfer continuity.
- Complex IV:Highly efficient and regulates oxygen use.
Regulation occurs through feedback mechanisms. Oxygen levels and energy demand affect component activity. These controls prevent excess reactive oxygen species and maintain balance.
Determining The Most Vital Component
Determining the most vital component in the electron transport chain (ETC) means looking beyond just names and functions. It requires understanding how each part contributes to the overall energy production in your cells. Have you ever wondered which component, if disrupted, would cause the biggest impact on your body’s ability to generate energy?
Criteria For Vitality
Vitality in the ETC is measured by a component’s role in electron transfer, proton pumping, and energy conversion efficiency. Components that directly drive the creation of the proton gradient tend to be more critical.
Consider these criteria:
- Electron transfer efficiency:How well does the component move electrons along the chain?
- Proton pumping ability:Does it contribute to building the proton gradient essential for ATP synthesis?
- Susceptibility to inhibition:How sensitive is the component to toxins or mutations?
Understanding these helps you see why some components are indispensable while others serve more supportive roles.
Impact On Cellular Respiration
Each component affects cellular respiration differently, but some have a larger effect on ATP production. For example, Complex IV (cytochrome c oxidase) completes the final step of electron transfer to oxygen. This makes it crucial because blocking it stops the entire chain.
On the other hand, if Complex II is impaired, the chain can still function through other pathways, though less efficiently. This shows how some components have backup systems, while others do not.
Ask yourself: What happens to your energy levels when a key component is damaged? Recognizing this can guide research or treatments targeting mitochondrial diseases.
Scientific Perspectives
The Electron Transport Chain (ETC) is a complex but vital process in cellular respiration. Understanding which component plays a crucial role can change how you view energy production in your body. Scientific perspectives offer valuable insights by combining detailed research and expert interpretations.
Research Findings
Scientists have pinpointed Complex IV, also known as cytochrome c oxidase, as a key player in the ETC. This component is responsible for the final transfer of electrons to oxygen, forming water. Without this step, the whole chain would back up, halting energy production.
Studies show that Complex IV’s efficiency directly affects how well cells generate ATP, the energy currency. Research involving mutations in this complex reveals how critical its function is—small changes can lead to serious metabolic diseases. Have you ever wondered why some people feel fatigued despite resting? It might be linked to subtle defects in this stage.
Expert Opinions
Leading biochemists emphasize the importance of Complex IV’s role in maintaining cellular health. Many agree that targeting this component can open new doors for treating mitochondrial disorders. Experts also highlight the adaptability of Complex IV under different oxygen levels, showing how your body adjusts to environmental changes.
From a practical viewpoint, researchers suggest that understanding this component’s function can help you optimize your health. For example, certain supplements and lifestyle choices may support the efficiency of Complex IV. What steps could you take today to support your cellular energy production?

Credit: echowater.com
Frequently Asked Questions
Which Component In The Electron Transport Chain Produces Atp?
ATP synthase is the key component that produces ATP. It uses the proton gradient to convert ADP and phosphate into ATP. This process is essential for cellular energy production.
What Is The Role Of Complex I In The Electron Transport Chain?
Complex I transfers electrons from NADH to ubiquinone. It also pumps protons into the intermembrane space, creating a proton gradient for ATP synthesis.
How Does Complex Iv Contribute To The Electron Transport Chain?
Complex IV transfers electrons to oxygen, the final electron acceptor. This forms water and helps maintain the proton gradient needed for ATP production.
Why Is Ubiquinone Important In The Electron Transport Chain?
Ubiquinone (coenzyme Q) transports electrons between Complexes I/II and III. It is lipid-soluble, allowing it to move freely within the inner mitochondrial membrane.
Conclusion
The electron transport chain has key parts that work together. Each component plays a role in making energy for cells. Understanding these parts helps explain how our bodies create power. The chain moves electrons and pumps protons to make ATP.
This process keeps living things alive and active. Knowing which component does what makes biology easier to grasp. Keep exploring to learn more about cell energy and life. Simple steps lead to big discoveries.
Leave a Reply